��Ŀ����

��֪��1.0mol/LFe��NO3��3��Һ��pH=1����ش��������⣺
��1��װ��A�з�����Ӧ�Ļ�ѧ����ʽ��
��2��Ϊ�ų�������ʵ��ĸ��ţ��μ�Ũ����֮ǰӦ���еIJ�����
��3��װ��B�в����˰�ɫ��������ɷ���
��4������B�в�����ɫ������ԭ��
����1��
����2��SO2��Fe3+��Ӧ��
����3��SO2�����������µ�N
| O | - 3 |
�ٰ�����2��װ��B�з�Ӧ�����ӷ���ʽ��
�ڰ�����3��ֻ�轫װ��B�е�Fe��NO3��3��Һ�滻�ɵ������������Һ������ͬ�����½���ʵ�飮Ӧѡ����Լ���
a.0.1mol/Lϡ����
b.1.5mol/L Fe��NO3��2��Һ
c.6.0mol/L NaNO3��Һ��0.2mol/L����������ϵ���Һ��
��2��Ϊ�ų�������ʵ��ĸ��ţ��μ�Ũ����֮ǰӦ��ͨ�뵪�����������ų���
��3������������л�ԭ�ԣ�������������ӷ���������ԭ��Ӧ������������ӣ������������ᱵ������
��4��Fe3+��Fe2+�����������ԣ��������������Ӧ���ɸ��ݷ�Ӧ��������жϣ�
�ʴ�Ϊ��Na2SO3+H2S04��Ũ��=Na2S04+SO2��+H2O��
��2��Ϊ�ų�������ʵ��ĸ��ţ��μ�Ũ����֮ǰӦ��ͨ�뵪�����������ų�������Ϊ���ɼУ���װ����ͨ��һ��ʱ���N2���رյ��ɼУ�
�ʴ�Ϊ�����ɼУ���װ����ͨ��һ��ʱ���N2���رյ��ɼУ�
��3������������л�ԭ�ԣ�������������ӷ���������ԭ��Ӧ������������ӣ������������ᱵ�������ʴ�Ϊ��BaS04����ԭ�ԣ�
��4������һ �������Ӷ�������������SO2��Fe3+������������NO3-����Ӧ���ʴ�Ϊ��SO2��Fe3+������������NO3-����Ӧ��
�ٰ��۵�2��װ��B�з�Ӧ�����ӷ���ʽ��SO2+2Fe3++Ba2++2H2O=BaSO4��+2Fe2++4H+����ͨ�������Ƿ���Fe2+���ɵķ����жϣ���������Ϊȡ����B����Һ���Թ��У������������軯��[K3Fe��CN��6]��Һ������ɫ����������Һ����Fe2+���۵�2������
�ʴ�Ϊ��SO2+2Fe3++Ba2++2H2O=BaSO4��+2Fe2++4H+��
�ڰ��۵�3��������������SO2��NO3-��Ӧ��ʵ����c��NO3-��=3mol/L�������6.0mol?L-1NaNO3��0.2mol?L-1����������ϵ���Һ���棬��ѡc��

 ���ɿ��õ�Ԫ����AB��ϵ�д�
���ɿ��õ�Ԫ����AB��ϵ�д� С�����ϵ�д�
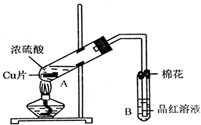
С�����ϵ�д�ij�о�С������ͼ��ʾװ�ý���ͭ��Ũ���ᷴӦ��ʵ���о���
��1��д���Թ�B�е�ʵ������ ��
��2��д��A�з�Ӧ�Ļ�ѧ����ʽ�� ��
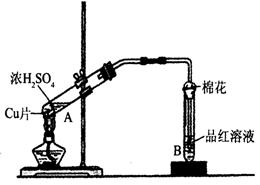 ��3����ַ�Ӧ����A�Թ�����ͭƬʣ�࣬������A�м���
��3����ַ�Ӧ����A�Թ�����ͭƬʣ�࣬������A�м���
NaNO3������ͭƬ�ܽ⣬��Ӧ�����ӷ���ʽ
Ϊ ��
��4����ַ�Ӧ����A�Թ�����ͭƬʣ�࣬
�����ְ�ɫ���ǣ��ð�ɫ������ ��
����ȷ�ϸð�ɫ������ʲô���ʵ�ʵ���������
�� ��
��5��B�Թܿڵ���Ӧմ�е��Լ��� ��
��6��С���Ա��4����Ӧ�����Һ�м�������������ͭ��ʹʣ�������ȫ��ת��Ϊ����ͭ�����˺���Һ����Ũ������ȴ��ᾧ�Ƶ�����ͭ���壨CuSO4��xH2O����С���Ա���ü��ȷ��ⶨ�þ�����ᾧˮx��ֵ
�������ǵ�ʵ������У����ٳ����ĴΣ�������γ�����Ŀ����
������������һ��ʵ������ݣ�
| �������� | �����뾧��������� | ���Ⱥ���������������� |
| 11.0g | 37.8g | 27.0g |
�����ϱ����ݼ����ж�x��ʵ��ֵ������ֵ��x��5�� ���ƫ����ƫС������
 ij�о�С������ͼ��ʾװ�ý���ͭ��Ũ���ᷴӦ��ʵ���о���
ij�о�С������ͼ��ʾװ�ý���ͭ��Ũ���ᷴӦ��ʵ���о���