��Ŀ����
ij��ҵ��ˮ�����±��е�ijЩ���ӣ��Ҹ������ӵ����ʵ���Ũ����ȣ���Ϊ0.1mol/L������ֵ����ˮ�ĵ��뼰���ӵ�ˮ�⣩��
| ������ | K+ Ag+ Mg2+ Cu2+ Al3+ |
| ������ | C1�� |
��ͬѧ��̽����ˮ����ɣ�����������ʵ�飺
��ȡ����ɫ��Һ5 mL���μ�һ�ΰ�ˮ�г������ɣ��������������ӡ�
���ò�˿պȡ��Һ���ڻ��������գ�����ɫ�ܲ����۲죬����ɫ���档
����ȡ��Һ����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ��
������������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɡ�
���ƶϣ�
��1����I�����жϣ���Һ��һ�������е��������� ��
��2�����м�������������ɫ��������ӷ���ʽ�� ��
��3����ͬѧ����ȷ��ԭ��Һ�������������� ���������� �����ݴ��Ʋ�ԭ��ҺӦ�ó� �ԣ�ԭ���� ���������ӷ���ʽ˵������
��4����ȡ100 mLԭ��Һ������������NaOH��Һ���˹������漰�����ӷ���ʽΪ
����ַ�Ӧ����ˣ�ϴ�ӣ����ճ��������أ��õ��Ĺ�������Ϊ g��
��֪ʶ�㡿���������ӵļ��顢���������ӵļ���
���𰸽�������1��K+��NH��Cu2+��2�֣�
������2��6I��+2NO+8H+===3I2+2NO��+4H2O��2�֣�
������3��Mg2+��Al3+��2�֣�����Cl����NO��SO ��I����3�֣������ᣨ1�֣�
��I����3�֣������ᣨ1�֣�
�������� Mg2++2H2O Mg(OH)2+2H+��Al3++3H2O
Mg(OH)2+2H+��Al3++3H2O Al(OH)3+3H+��1�֣�д������һ�����ɣ�
Al(OH)3+3H+��1�֣�д������һ�����ɣ�
������4��Mg2++2OH��===Mg(OH)2����Al3++4OH��===AlO+2H2O��2�֣����������Ҳ�ɣ�����
�������� 0.4��1�֣�
��������ȡ����ɫ��Һ5mL��˵��һ��������Cu2+���μ�һ�ΰ�ˮ�г������ɣ��������������ӣ�˵�����ӵ���NH4+������ԭ��Һ��һ������NH4+�����ܺ���Mg2+��Al3+������NH4+��CO32-��
���ò�˿պȡ������Һ���ڻ��������գ�����ɫ���棨����ɫ�ܲ����۲죩��˵��û��K+��
����ȡ��Һ�����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ����ʱ��Һ��Ȼ���壬˵���л�ԭ������ I-��NO3-��H+��Ӧ����NO������Һ����I-��NO3-�����ж�һ��������Ag+��
������������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɣ�˵����SO42-��
��1���ɢ��жϣ���Һ��һ�������е���������K+��NH4+��Cu2+��
��2�����м�����������������ɫ���壬��I-��NO3-��H+��Ӧ����NO�������ӷ���ʽ��6I-+2NO3-+8H+�T3I2+2NO��+4H2O��
��3��������������֪һ�����е�������I-��NO3-��SO42-���Ҹ�Ϊ0.1mol/L�����ݵ���غ��֪���ƶϳ��������Ӻ���Mg2+��Al3+����Ũ��Ϊ0.1mol/L���ɵ���غ��֪��Һ�л���һ��-1�۵�������ΪCl-�����Լ�ͬѧ����ȷ��ԭ��Һ�������������ǣ�Mg2+��Al3+���������ǣ�Cl-��I-��NO3-��SO42-����Һ��þ���Ӻ�������ˮ����Һ�����ԣ���Ӧ�����ӷ���ʽΪ��Mg2++2H2O⇌Mg��OH��2+2H+��Al3++3H2O⇌Al��OH��3+3H+��
��4����ȡ100mLԭ��Һ������������NaOH��Һ��Mg2+��Al3+ ��Ӧ����Mg��OH��2��NaAlO2���漰�����ӷ���ʽΪMg2++2OH-�TMg��OH��2����Al3++4OH-�TAlO2-+2H2O����ַ�Ӧ����ˣ�ϴ�ӣ����ճ��������أ�����Ԫ���غ㣺n��MgO��=n��Mg2+��=cV=0.1mol/L��0.1L=0.01mol��n��MgO��=0.01mol��40g/mol=0.4g��
��˼·�㲦�����⿼�����ʵļ��顢�����Լ�����ʽ���йؼ��㣬��Ŀ�Ѷ��еȣ������Ĺؼ��ǰ����йط�Ӧ�Ļ�ѧ����ʽ����д��

��ͼ��ij��Һϡ�����У���Һ�����ʵ���Ũ������Һ����ı仯����ͼ������ͼ�����ݷ����ó�aֵ���ڣ� ����
A��2 B��3 C��4 D��5

�������ʷ������ȷ����ǣ� ��
| �������� ��ϡ��� | �� | �� | �� | ���������� | ���������� |
| A | Na2CO3 | H2SO4 | NaHCO3 | SiO2 | CO2 |
| B | NaOH | HCl | NaCl | Na2O | CO |
| C | NaOH | CH3COOH | MgCl2 | CO2 | SO2 |
| D | KOH | HNO3 | CaCO3 | CaO | SO3 |
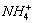



 I��
I�� B��
B�� C��
C��  D��
D��


 2HI(g)����һ���¶�����H2(g)��I2(g)��ʼ��Ӧ������˵����ȷ����(����)
2HI(g)����һ���¶�����H2(g)��I2(g)��ʼ��Ӧ������˵����ȷ����(����)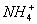 ��H����
��H����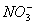 ��
��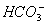
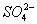 ��NH3��H2O
��NH3��H2O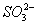 ��Cl2
��Cl2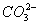 ��
��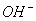
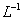 ������ζ�0.12 mol•.
������ζ�0.12 mol•.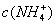 >
>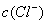 ,
,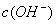 >
>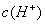 B.
B.