��Ŀ����
����Ŀ�����з�Ӧ�����ڼ��˾���Ƿ�ƺ��ʻ��
2Cr2O![]() ��3CH3CH2OH��16H����13H2O�D��4[Cr(H2O)6]3����3CH3COOH
��3CH3CH2OH��16H����13H2O�D��4[Cr(H2O)6]3����3CH3COOH
(1)Cr3����̬��������Ų�ʽΪ________�������[Cr(H2O)6]3���У���Cr3���γ���λ����ԭ����______(��Ԫ�ط���)��
(2)CH3COOH��Cԭ�ӹ���ӻ����ͷֱ�Ϊ____________��1 mol CH3COOH�����к��ЦҼ�����ĿΪ__________��
(3)��H2O��Ϊ�ȵ������һ��������Ϊ________(�ѧʽ)��
���𰸡� 1s22s22p63s23p63d3��[Ar]3d3 O sp3��sp2 7 mol(��7��6.02��1023) H2F��
�������� (1)Cr��24��Ԫ�أ������Ų�ʽΪ1s22s22p63s23p63d44s2����Cr3���ĺ�������Ų�ʽΪ1s22s22p63s23p63d3��[Ar]3d3��H2O�����е�Oԭ���й¶Ե��ӣ�����Cr3���γ���λ����(2)CH3COOH�м��ϵ�̼ԭ�Ӳ���sp3�ӻ����Ȼ���̼ԭ�Ӳ���sp2�ӻ���CH3COOH�ĽṹʽΪ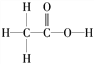 ��̼��˫���к���1������������ĵ�����������������7��������1 mol CH3COOH�����к��ЦҼ�����ĿΪ7 mol��7��6.02��1023��(3)H2O��������3��ԭ�Ӻ�8���۵��ӣ����以Ϊ�ȵ������������ΪH2F����
��̼��˫���к���1������������ĵ�����������������7��������1 mol CH3COOH�����к��ЦҼ�����ĿΪ7 mol��7��6.02��1023��(3)H2O��������3��ԭ�Ӻ�8���۵��ӣ����以Ϊ�ȵ������������ΪH2F����

����Ŀ��þ��ͭ�Ƚ��������������ڶ���ø�ĸ����ӡ���ҵ�ϴӺ�ˮ����ȡþʱ�����Ʊ���ˮ�Ȼ�þ��Ȼ�������ڵ�⣬�õ�����þ��
(1)��MgCl2Ϊԭ���������ε�ⷨ�Ʊ�þʱ��������NaCl��KCl��CaCl2�Ƚ����Ȼ������Ҫ���ó��˽����۵�֮���________��
(2)��֪MgO�ľ���ṹ����NaCl�͡�ijͬѧ������MgO�����ṹʾ��ͼ��ͼ��ʾ�������ͼ�д���__________________��

(3)Mg�ǵ�������Ԫ�أ������ڲ���Ԫ�ط�������۵���±���
������ | NaF | MgF2 | SiF4 |
�۵�/K | 1 266 | 1 534 | 183 |
���ͱ��з������۵�����ԭ��_____________________________________��