��Ŀ����
CO��H2������Ҫ�Ļ���ԭ�ϣ�CO2�Ĺ̶������öԻ�����������Դ����������Ҫ�����塣
��1������ˮú����CO+H2����Ϊ�ϳ�������ͬһ�����У�ѡ��˫��������������������Ӧ�����պϳɶ����ѣ�CH3OCH3����
�״��ϳɷ�Ӧ:CO(g)+2H2(g) CH3OH(g) ��H1=��90.8kJ��mol��1
CH3OH(g) ��H1=��90.8kJ��mol��1
ˮú���任��Ӧ��CO(g)+H2O(g) CO2(g)+H2(g) ��H2=��41.3kJ��mol��1
CO2(g)+H2(g) ��H2=��41.3kJ��mol��1
�״���ˮ��Ӧ��2CH3OH(g) CH3OCH3(g)+H2O(g) ��H3=��23.5kJ��mol��1
CH3OCH3(g)+H2O(g) ��H3=��23.5kJ��mol��1
д����CO��H2�ϳɶ����������ˮ�������Ȼ�ѧ����ʽ____________��
��2����2L�����ܱ������У�Ͷ��CO(g)��H2(g)��5mol���������·�Ӧ��
3CO(g)+3H2(g) CH3OCH3(g)+CO2(g) ��H��0��CO��ת���ʦ����¶ȡ�ѹǿ�Ĺ�ϵ��ͼ��ʾ��
CH3OCH3(g)+CO2(g) ��H��0��CO��ת���ʦ����¶ȡ�ѹǿ�Ĺ�ϵ��ͼ��ʾ��

��p1��p2��p3��������________��
�����÷�Ӧ����50minʱ�ﵽƽ�⣬��ʱCO��ת���ʦ�=0.6����50min��H2�ķ�Ӧ����Ϊ____mol��L��1��min��1��
��3�������ѣ�CH3OCH3��ȼ�ϵ�ؾ��������졣Ч�ʸߵ��ŵ㣬�������ܶȸ��ڼ״�ȼ�ϵ�ء��乤��ԭ����ͼ��ʾ��X������������______���������������ǿ�����䡱����Y�缫��_____����д��X�缫�Ϸ����ĵ缫��Ӧʽ__________�����øö�����ȼ�ϵ�ص�ⱥ��ʳ��ˮ������Ϊʯī�缫��������2.3gȼ�ϱ�����ʱ������������������Ϊ_____L����״���£�

 ��ҵ����ϵ�д�
��ҵ����ϵ�д� ͬ��ѧ��һ�ζ���ϵ�д�
ͬ��ѧ��һ�ζ���ϵ�д� �����ܾ�ϵ�д�
�����ܾ�ϵ�д���֪25�桢101kPaʱ��һЩ���ʵ�ȼ����Ϊ��
��ѧʽ | CO(g) | H2(g) | CH3OH(l) | CH4(g) |
��H/(kJ/mol) | -283.0 | -285.8 | -726.51 | -890. 31 |
��ش��������⣺
��1�����ݸ�˹����������з�Ӧ���Ȼ�ѧ����ʽ��
CO(g)+2H2(g)==CH3OH(l) ��H=_________��
��2������H2��CH4�Ļ������112 L����״������ʹ����ȫȼ������CO2(g)��H2O(l)�����ų�����3 242.5 kJ����ԭ���������H2��CH4�����ʵ���֮���ǣ�____________��
A.1:1 B.1:3 C.1:4 D.2:3
��һ�����İ����������ں��ݵ��ܱ���������У�����������Բ��ƣ���ʹ��ﵽ��ѧƽ�⣺H2NCOONH4(s) 2NH3(g)+CO2(g)��ʵ���ò�ͬ�¶��µ�ƽ�����������±�
2NH3(g)+CO2(g)��ʵ���ò�ͬ�¶��µ�ƽ�����������±�
�¶�/�� | 15.0 | 20.0 | 25.0 | 30.0 | 35.0 |
ƽ����ѹǿ/kPa | 5.7 | 8.3 | 12.0 | 17.1 | 24.0 |
ƽ��������Ũ��/10��3mol/L | 2.4 | 3.4 | 4.8 | 6.8 | 9.4 |
�����й�������ȷ����
A. �ڵ����¸÷�Ӧ���Է�����
B. 15��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��ԼΪ2.0
C. ���������ƽ����Է�����������ʱ���÷�Ӧ�ﵽ��ѧ��Ӧ��
D. ���������£���ԭƽ����ϵ���ٳ���2molNH3��1molCO2����ƽ���CO2Ũ�Ȳ���
��һ������SO2ͨ��FeCl3��Һ�У�ȡ�����Һ���ֱ��������ʵ�飬��֤��SO2��FeCl3��Һ����������ԭ��Ӧ����
���� | ���� | |
A | ����NaOH��Һ | �к��ɫ���� |
B | ����Ba(NO3)2��Һ | �а�ɫ���� |
C | ��������KMnO4��Һ | ��ɫ��ȥ |
D | ����K3[Fe(CN)6]�����軯�أ���Һ | ����ɫ���� |
A. A B. B C. C D. D
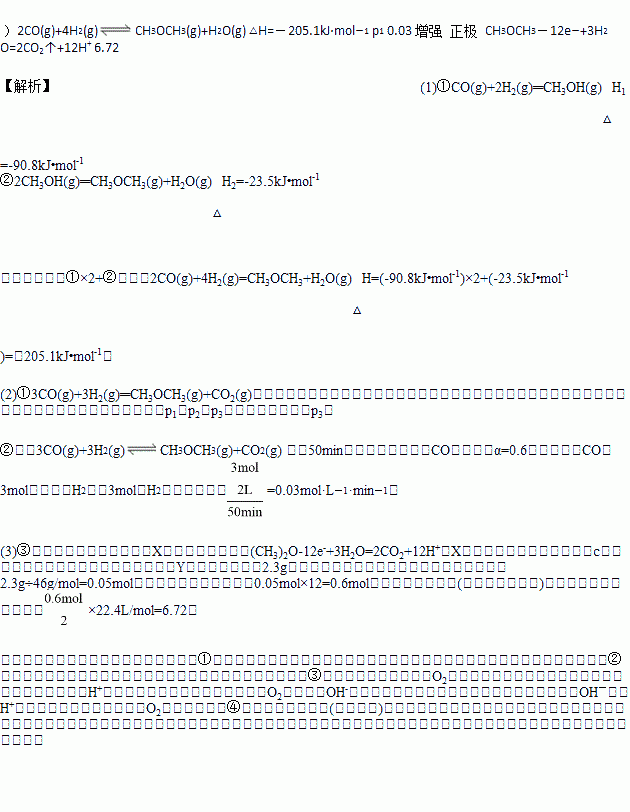

 pC(g)+qD(g)����H���ڲ�ͬ�¶��¾���һ��ʱ����������ϵ��C�İٷֺ������¶�T�Ĺ�ϵ��ͼ����ʾ��һ�������´ﵽƽ���tʱ�̸ı�Ӱ��ƽ�����һ�����������½���ƽ��ķ�Ӧ���̡��ɴ˿��жϸ÷�Ӧ��
pC(g)+qD(g)����H���ڲ�ͬ�¶��¾���һ��ʱ����������ϵ��C�İٷֺ������¶�T�Ĺ�ϵ��ͼ����ʾ��һ�������´ﵽƽ���tʱ�̸ı�Ӱ��ƽ�����һ�����������½���ƽ��ķ�Ӧ���̡��ɴ˿��жϸ÷�Ӧ��
 ��0
��0 ���������ɲ�ȡ�Ĵ�ʩ��( )
���������ɲ�ȡ�Ĵ�ʩ��( ) ���� B. ��������
���� B. �������� ����
����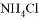 ���� D. ��������
���� D. ��������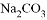 ����
����