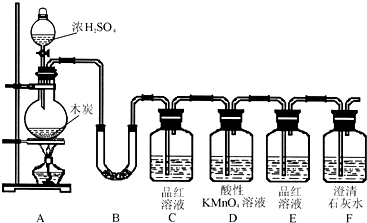
��Ŀ����
11����6ƿ��ɫ�����Լ����ֱ������������������ơ������ơ�����李���ˮ����ͭ��̼���ƣ���ֻ�ṩ����ˮ��ͨ�������ʵ�鲽�輴��ǩ�����ǣ�����д���пհף���1����ȡ���������Լ��ֱ����6֧�Ժ��У�������������ˮ�����Թܣ��۲쵽�����������ֹ���ȫ���ܽ⣬��������Ϊ��ɫ��Һ��ֻ��һ������ɫ��Һ��
����������ʵĻ�ѧʽ������ʽ����CuSO4��
��2���ֱ�ȡδ�������Һ�������м��������Ѽ������Һ���۲쵽���������£������������������д����Ӧ�����ӷ���ʽ��
����1֧�Թ����а�ɫ��������Ba2++SO42-�TBaSO4����
����2֧�Թ�������ɫ������Cu2++2OH-�TCu ��OH��2����Cu2++CO32-�TCuCO3����˳����Ի�������
����������ʵĻ�ѧʽ������ʽ����BaCl2��
��3����������δ������ʵķ����۲쵽������ֱ��ǣ��ٷֱ�ȡ��2�����ܹ�������ɫ������������Һ�������м���BaCl2��Һ���а�ɫ�������ɵ���Na2CO3��û�г�����������NaOH���ڷֱ�ȡ����δ�������Һ�������м���NaOH��Һ���������������Na2SO4���д̼�����ζ����������ǣ�NH4��2SO4��
���� ��1����������ε��ܽ��Կ���֪���������������ʾ��ܹ�����ˮ����������ͭ����ˮ���õ���ɫ��Һ��
��2���ܹ�������ͭ��Ӧ���������Ȼ������������ơ�̼���Ƶ��������ʣ��ʿ���д�����Ƿ�Ӧ�����ӷ���ʽ��
��3�����ݣ�1����2����ʵ����������ͭ���Ȼ���������������������Ѽ�����������ʺ�ʣ�����������֮����Խ��������ֿ�����
��� �⣺��1����ˮ����ͭ����ˮ����Һ��Ϊ��ɫ���������������������ܽ��õ�������ɫ��Һ����������ͭ�������ȱ����������
�ʴ�Ϊ�����ֹ���ȫ���ܽ⣬��������Ϊ��ɫ��Һ��ֻ��һ������ɫ��Һ��CuSO4��
��2�����Ȼ������Ժ�����ͭ��Ӧ���ɰ�ɫ�����ᱵ���������ӷ���ʽΪBa2++SO42-=BaSO4�����ʴ�Ϊ��Ba2++SO42-=BaSO4����
������ͭ������������Һ��Ӧ����������ͭ��ɫ���������ӷ���ʽΪCu2++2OH-=Cu��OH��2������
����ͭ��̼������Һ��Ӧ������ɫ̼��ͭ��������Ӧ�����ӷ���ʽ2Cu2++2CO32-+H2O=Cu2��OH��2CO3��+CO2����
�����Ȼ������Ա����������
�ʴ�Ϊ��Cu2++2OH-=Cu��OH��2����2Cu2++2CO32-+H2O=Cu2��OH��2CO3��+CO2����BaCl2��
��3���ֱ�ȡ��2�����ܲ�����ɫ������������Һ�������м���BaCl2��Һ���а�ɫ�������ɵ���Na2CO3��Һ���������ɵ���NaOH��Һ���ֱ�ȡ����δ�������Һ�������м���NaOH��Һ���������������Na2SO4��Һ���д̼�����ζ����������ǣ�NH4��2SO4��Һ��
�ʴ�Ϊ���ٷֱ�ȡ��2�����ܹ�������ɫ������������Һ�������м���BaCl2��Һ���а�ɫ�������ɵ���Na2CO3��û�г�����������NaOH�� �ڷֱ�ȡ����δ�������Һ�������м���NaOH��Һ���������������Na2SO4���д̼�����ζ����������ǣ�NH4��2SO4��
���� ��ɫ����ˮ����ͭ����ˮ��õ���ɫ��Һ�ǽ���ؼ���Ȼ������������֮��ķ�Ӧ������һһ����

| A�� | ʵ���������Ľ����Ʊ�����ú�ͻ�ʯ������ | |
| B�� | �������Ż��ø����ɳ������� | |
| C�� | ���Ʋ;߲������������ʱ�������ԡ����Ի��̵�ʳ�� | |
| D�� | ���ƴ��߲��������峴ʳ���Ϊ��Ҫ��ˮ��Ӧ |
| A�� | 2V��������N2��=V���棩��NH3�� | B�� | V��������N2��=3V��������H2�� | ||
| C�� | 2V��������H2��=3V���棩��NH3�� | D�� | N2��H2��NH3�������Ϊ1��3��2 |
| A�� | 1��2 | B�� | 2��1 | C�� | 1��3 | D�� | 3��1 |
| A�� | 16g CH4��18 g NH4+ ������������� | |
| B�� | 1molNa2O2������ˮ��Ӧת��2NA������ | |
| C�� | ������2.24 L CO2�к��е�ԭ����Ϊ0.3��6.02��1023 | |
| D�� | NA��һ����̼���Ӻ�0.5 mol�����������Ϊ7��4 |
| A�� | ʹ��Cu-Zn-Fe�����ɴ���������Ч�� | |
| B�� | �����¶ȣ��÷�Ӧƽ�ⳣ��Kһ������ | |
| C�� | �������CO2��������H2��ת���� | |
| D�� | ��ƽ���������з����CH3CH2OH��H2O�������CO2��H2�������� |