��Ŀ����
����Ŀ����ҵ�ϵ�ⷨ�����������Է�ˮ���õ�����Ni��ԭ����ͼ��ʾ������˵������ȷ��
�ǣ� ��

��֪���ŵ�˳��Ni2+(��Ũ��)>H+> Ni2+(��Ũ��)
A. ̼���Ϸ����ĵ缫��Ӧ��4OH--4e-�TO2��+2H2O
B. Ϊ�����Ni�IJ��ʣ�����������Ҫ���Ʒ�ˮpH
C. �������У�B��NaCl��Һ�����ʵ���Ũ�Ƚ���������
D. ����ͼ��������Ĥȥ������A��B���Һϲ������ⷴӦ�ܷ���ʽ���ᷢ���ı�
���𰸡�D
��������A����ͼ֪��̼�����Դ���������ǵ��ص��������缫��Ӧ4OH--4e-=2H2O+O2������A��ȷ��B����Ni2+����������Һ������ˮ�⣻�����ԣ�Ni2+(��Ũ��)��H+��Ni2+(��Ũ��)��Ϊ�����Ni�IJ��ʣ�����������Ҫ���Ʒ�ˮpH����B��ȷ��C�������������Դ���������ǵ��ص��������缫��ӦNi2++2e-=Ni����������Ϊƽ��A��C�еĵ�ɣ�A�е�Na+��C�е�Cl-�ֱ�ͨ��������Ĥ��������Ĥ����B�У���ʹB��NaCl��Һ�����ʵ���Ũ�Ȳ�������C��ȷ��D������ͼ��������Ĥȥ�������ڷŵ�˳��Cl-��OH-����Cl-���������ŵ磺2Cl---2e-=Cl2������ⷴӦ�ܷ���ʽ�ᷢ���ı䣬��D����ѡD��

����Ŀ��A��B��C��D��E�� F��G�����ڱ��ж����ڵ�����Ԫ�أ��й����ʻ�ṹ��Ϣ���±���
Ԫ�� | �й����ʻ�ṹ��Ϣ |
A | �ؿ��к�������Ԫ�� |
B | B��������A�����ӵ�������ͬ���������������е������Ӱ뾶��С�� |
C | C��Bͬ���ڣ���������������ԭ�Ӱ뾶����(ϡ���������) |
D | Dԭ�������������ǵ��Ӳ�����2�������⻯���г�������ζ |
E | E��Dͬ���ڣ����ڸ�������ԭ�Ӱ뾶��С |
F | F���⻯�������������ˮ���ﷴӦ����һ�����ӻ����� |
G | G���γɻ�������������Ԫ�� |
(1) BԪ�ط���Ϊ____��A��C��ԭ�Ӹ�����Ϊ1��1�γɵĻ�����ĵ���ʽΪ___���õ���ʽ��ʾC��E�γɻ�����Ĺ���____�� D�γɵļ����ӵĽṹʾ��ͼΪ____��
(2) F���⻯������____(���Ի�Ǽ���)���γɵķ��ӣ�д��ʵ�����Ʊ����⻯��Ļ�ѧ����ʽ____��
(3) �ǽ�����D____E(����ڻ�С��)�����ԭ�ӽṹ�ĽǶȽ���ԭ��__��
����Ŀ����1��CH3COOH��������л����ᡣ
��CH3COOH�ĵ��뷽��ʽΪ_____________��
��CH3COONa��Һ�ʼ��ԣ�ԭ����__________������CH3COO-������Na+����ˮ���������
�� ��CH3COONa��Һ����c(Na+)_______c(CH3COO-)��������������=������������
��2����֪����ӦC(s)+H2O(g)![]() CO(g)+H2(g)����H��131.3kJ/mol
CO(g)+H2(g)����H��131.3kJ/mol
�� �÷�Ӧ�Ƿ�Ӧ__________��������������������������
�� ����Ӧ����24gC(s)����Ӧ����ЧӦΪ__________��
��3����һ���¶��£���CO(g)��H2O(g)��0.16 mol ͨ���ݻ�Ϊ2.0L�ĺ����ܱ����������������·�Ӧ��CO(g)+H2O(g) ![]() CO2(g)+H2(g)���õ��������ݣ�
CO2(g)+H2(g)���õ��������ݣ�
t/min | 2 | 4 | 7 | 9 |
n(H2O)/mol | 0.12 | 0.11 | 0.10 | 0.10 |
�� 2 min �ڣ�vCO=__________��
�� ���¶��£��˷�Ӧ��ƽ�ⳣ������ʽK=_________��
�� �����������䣬�ٳ���0.1molCO��0.1molH2O(g)���ﵽƽ��ʱCO ���ݻ�����_____������������������С����������������
��4����ͼΪֱ����Դ���CuSO4��Һ��װ�ã�C1��C2��Ϊʯī�缫����ش��������⣺
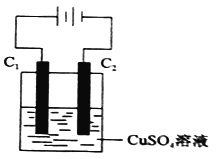
�� C1��_________��������������������������
�� C2���缫��ӦʽΪ____________��

