��Ŀ����
14��̼���仯����㷺��������Ȼ���У��ش��������⣺��1������һ���ռ��˶�״̬�ĵ�����ԭ�Ӻ�����ֵĸ����ܶȷֲ����õ����������������ڻ�̬14Cԭ���У��������2�������෴�ĵ��ӣ�
��2��CS2�����У����ۼ��������ЦҼ��ͦ�����
��3��CO�������Fe�γ�Fe��CO��5���û������۵�Ϊ253K���е�Ϊ376K����������ڷ��Ӿ��壮
��4��̼�ж���ͬ�������壬����ʯīϩ����ʯ�ľ���ṹ��ͼ��ʾ��
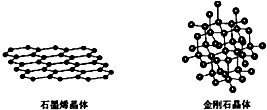
����ʯīϩ�����У�ÿ��Cԭ������3����Ԫ����ÿ����Ԫ��ռ��2��Cԭ�ӣ�
���ڽ��ʯ�����У�Cԭ�������ӵ���С��ҲΪ��Ԫ����ÿ��Cԭ������12����Ԫ����
���� ��1�������Ʊ�ʾ������ԭ�Ӻ�����ֻ���Ķ��٣��ڻ�̬14Cԭ���У���2�ԳɶԵ��ӣ��ɶԵ����������������������෴��
��2��CS2���ӽṹʽΪS=C=S�����۵���Ϊ�Ҽ�������˫����һ���ǦҼ���һ���Ǧ�����
��3�����Ӿ�����۷е�ϵͣ�
��4����ÿ����Ԫ��ֻ��Cԭ�Ӹ���=$\frac{1}{3}��6$��
���ڽ��ʯ�����У�Cԭ�������ӵ���С��Ϊ��Ԫ����ÿ��Cԭ������4��̼ԭ�ӣ����ӵ�4��Cԭ����ÿ��Cԭ���γ�3����Ԫ����
��� �⣺��1�������Ʊ�ʾ������ԭ�Ӻ�����ֻ���Ķ��٣��ڻ�̬14Cԭ���У���2�ԳɶԵ��ӣ��ɶԵ����������������������෴�������ڻ�̬14Cԭ���У������������������෴�ĵ��ӣ��ʴ�Ϊ�������ƣ�2��
��2��CS2���ӽṹʽΪS=C=S�����۵���Ϊ�Ҽ�������˫����һ���ǦҼ���һ���Ǧ����������ڶ���̼�д��ڦҼ��ͦ������ʴ�Ϊ���Ҽ��ͦ�����
��3�����Ӿ�����۷е�ϵͣ��û������۵�Ϊ253K���е�Ϊ376K���������ڷ��Ӿ��壬�ʴ�Ϊ�����ӣ�
��4����ÿ����Ԫ��ֻ��Cԭ�Ӹ���=$\frac{1}{3}��6$=2���ʴ�Ϊ��2��
���ڽ��ʯ�����У�Cԭ�������ӵ���С��Ϊ��Ԫ����ÿ��Cԭ������4��̼ԭ�ӣ����ӵ�4��Cԭ����ÿ��Cԭ���γ�3����Ԫ�������Խ��ʯ������ÿ��Cԭ�����γ�12����Ԫ�����ʴ�Ϊ��12��
���� ���⿼�����ʽṹ�����ʣ�Ϊ��Ƶ���㣬�漰���ʯ�ṹ�����������жϡ���ѧ����ԭ�Ӻ�������Ų���֪ʶ�㣬���ؿ���ѧ�������жϼ��ռ������������ѵ��ǣ�4���Тڣ���Ϥ���ʯ�ṹ�ص㼴�ɽ����Ŀ�Ѷ��еȣ�

 ����ν����Ž̲��㽭���̴�ѧ������ϵ�д�
����ν����Ž̲��㽭���̴�ѧ������ϵ�д� �����Ļ������������������ϵ�д�
�����Ļ������������������ϵ�д�| A�� | ����ͭ�����ڶ�����Ԫ�� | |
| B�� | CuO��Al2O3�����ڼ��������� | |
| C�� | ���ģ���������õ����ޣ�ͭ������ | |
| D�� | Ŀǰ��ҵ�ϻ��ͭ�����������Ȼ�ԭ�� |
| A�� | ������Ϊ78 | B�� | ������Ϊ53 | C�� | ������Ϊ131 | D�� | ���������Ϊ78 |
| A�� | ����ˮ������Al��OH��3���壬������ˮ�� | |
| B�� | SO2����Ư���ԣ�������Ư��ֽ�� | |
| C�� | ̼������Һ�Լ��ԣ������ȵĴ�����Һ��ȥ���������������� | |
| D�� | ������ѧ����ͨ�������ã��ɽ����ȵ�þ�ۿɷ��ڵ�������ȴ |
| A�� | H+H��H-H | |
| B�� | 2NH4Cl+Ba��OH��2•8H2O�TBaCl2+2NH3��+10H2O | |
| C�� | Mg+2HCl�TMgCl2+H2�� | |
| D�� | H2SO4+2KOH�TK2SO4+2H2O |
| A�� | ���³�ѹ�£�15g��������к��е�̼�����ĿΪ3NA | |
| B�� | ��״̬�£�22.4L�ı��ķ�����ΪNA | |
| C�� | ͨ��״̬�£�1.7g��NH3�к���������ΪNA | |
| D�� | 23gNa����ˮ�з�Ӧ��ʧȥ����2NA |
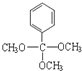 ��
��
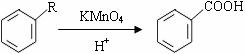
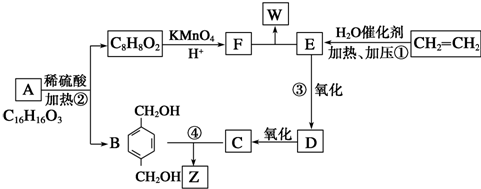
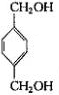 +2CH3COOH$��_{��}^{Ũ����}$
+2CH3COOH$��_{��}^{Ũ����}$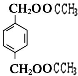 +2H2O��
+2H2O�� ��
��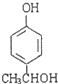 ��
��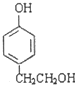 ��
��