��Ŀ����
ij��ѧС������Ũ����Ͷ��������ڼ�����������ȡ���������������������йص�̽��ʵ�飬��ȡ������װ����ͼI�͢�
��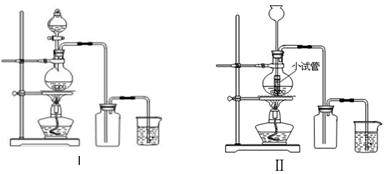
��1����ȡ�����ķ�Ӧ�����ӷ���ʽ ��
��2��װ�â���С�Թܵ�����Ϊ ��
��3��װ��I��װ�â�Ƚϣ�װ��I����Ҫ�ŵ�Ϊ�� ��
��4�������ɵ�����ͨ��ˮ�У����Ƶ���ˮ����ˮ�����ֽ����� ���塣
��5��������ʹʪ��ĺ�ɫ������ɫ������ʹ��ɫ������ɫ�����ʣ�ͬѧ�ǵĿ�����һ�£���Ϊ��ˮ�д��ڵļ������Ӷ��п��ܣ���������ѧʵ���ҳ����Լ������ʵ�飬�ó���ȷ���ۡ�
| ������� | �ռ����� | ������� | ��֤���� | �ó����� |
| ��ˮ�к���������ʹʪ��ĺ�ɫ������ɫ�� | ��Cl2��ǿ������ ��Cl2����ˮ��Ӧ���������HClO ��HClO��ǿ������ | �� �� ������ʹ������ɫ�� �� �� ��H2Oʹ������ɫ | ��֤����٣��Ѻ�ɫ�ɲ��������Cl2�ļ���ƿ����������ɫ�� ��֤����ڣ� �� ��֤����ܣ��Ѻ�ɫ��������ˮ���������ɫ�� | ʹ��ɫ������ɫ�������� �� |
��1��MnO2 �� 4H+ �� 2Cl��![]() Mn2+ �� 2H2O �� Cl2 �� ��2�֣�
Mn2+ �� 2H2O �� Cl2 �� ��2�֣�
��2��Һ����ֹ������� ��2�֣�
��3�����ڿ��Ƶμ�Ũ���ᣬ����Ũ����Ļӷ� �������� ��2�֣�
��4������ ��2�֣�
��5������ʹ������ɫ ��2�֣� HClOʹ������ɫ ��2�֣�
�Ѻ�ɫ��������������Һ�ﲻ��ɫ ��2�֣� HClO ��2�֣�

ij��ѧС������Ũ����Ͷ��������ڼ�����������ȡ���������������������йص�̽��ʵ�� ����ȡ������װ����ͼI�͢�
����ȡ������װ����ͼI�͢� ��
��
��1����ȡ�����ķ�Ӧ�����ӷ���ʽ ��
��2��װ�â���С�Թܵ�����Ϊ ��
��3��װ��I��װ�â�Ƚϣ�װ��I����Ҫ�ŵ�Ϊ�� ��
��4�������ɵ�����ͨ��ˮ�У����Ƶ���ˮ����ˮ�����ֽ����� ���塣
��5��������ʹʪ��ĺ�ɫ������ɫ������ʹ��ɫ������ɫ�����ʣ�ͬѧ�ǵĿ�����һ�£���Ϊ��ˮ�д��ڵļ������Ӷ��п��ܣ���������ѧʵ���ҳ����Լ������ʵ�飬�ó���ȷ���ۡ�
| ������� | �ռ����� | ������� | ��֤���� | �ó����� |
| ��ˮ�к���������ʹʪ��ĺ�ɫ������ɫ�� | ��Cl2��ǿ������ ��Cl2����ˮ��Ӧ���������HClO ��HClO��ǿ������ | �� �� ������ʹ������ɫ�� �� �� ��H2Oʹ������ɫ | ��֤����٣��Ѻ�ɫ�ɲ��������Cl2�ļ���ƿ����������ɫ�� ��֤����ڣ� �� ��֤����ܣ��Ѻ�ɫ��������ˮ���������ɫ�� | ʹ��ɫ������ɫ�������� �� |
 H++HCO3- Ka1 =4��45��10-7
H++HCO3- Ka1 =4��45��10-7
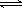 H++HCO3-
Ka1 =4��45��10-7
H++HCO3-
Ka1 =4��45��10-7