��Ŀ����
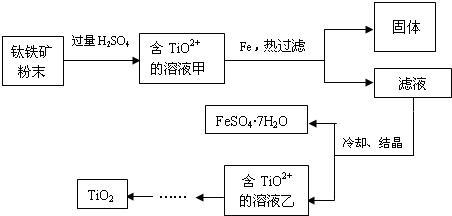
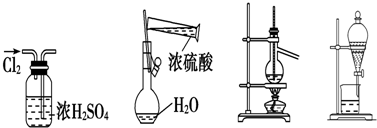
7������ͭ��һ��Ӧ�ü���㷺�Ļ���ԭ�ϣ����ò�ͬ�ķ����Ƶ�����ͭ��������Ũ����ֶ�μӵ�ͭ����ϡ����Ļ�����У�����ʹ֮��Ӧ��ȫ��ͨ���������ᾧ�õ�����ͭ���壨װ����ͼ1��2��ʾ����������ͭ������С���������ȼ�����ˮ����ͭ��
��1��ͼ1��Һ©����װ��Һ����Ũ���ᣮͼ2��ͼ1�ĸĽ�װ�ã���ͼ1��ȣ�ͼ2װ�õ������ŵ��Ƿ�ֹ�������к������ܱ���ȫ���գ�
��2�����Ƶõ���ˮ����ͭ����ij˫��ˮ���Ƿ�ˮʱ�����˷��ֹ�������⣬�����ָ�˫��ˮ�������ݲ������Դ����кβ���ͭ���Ӷ�˫��ˮ�ֽ��д����ã���
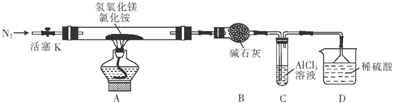
��ʵ������Ũ������ͭ��Ӧ���Խ�һ����ȡ����NaHSO3��ʵ��װ����ͼ3��ʾ��
��ش�
��3��ʵ����ȡһ����CuƬ��һ����ŨH2SO4����Բ����ƿ�й��ȣ�����Ӧ��������Ӧ�����Һ�м���������CuO�����˺���Һ����Ũ������ȴ�ᾧ�Ƶ�����ͭ���壨CuSO4•xH2O����ijС��ͬѧ���ü��ȷ��ⶨ�þ�����ᾧˮx��ֵ��
����д��װ�ü��з�����Ӧ�Ļ�ѧ����ʽCu+2H2SO4$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��
�ڼ��ȷ��ⶨ�þ�����ᾧˮx��ֵ���ᾧˮ��ȫʧȥ���жϷ��������γ���������0.1g��
�����������ʵ�����ݵ�ƽ��ֵ
| �������� | �����뾧�������� | ���Ⱥ���������������� |
| 11.7g | 23.3g | 19.7g |
��4������˵����ȷ����bd������ţ���
a����װ��ʹ�õIJ��������У��ƾ��ơ������ܡ�����©����Բ����ƿ
b��KMnO4��Һ����β������
c������Ʒ����Һ���뵽��ƿ�У���Ʒ�첻��ɫ��˵����NaHSO3����
d������Ʒ����Һ���뵽��ƿ�У���Ʒ����ɫ��˵��NaOH����ȫת��ΪNaHSO3
e������Ʒ����Һ�������Ը��������Һ�������뵽��ƿ�У������Ϻ�ɫ��˵��NaOH����ȫת��ΪNaHSO3

���� ��1���������⽫����Ũ����ֶ�μӵ�ͭ����ϡ����Ļ�����У���Һ©����ӦΪŨ���ᣬŨ���ᱻ��ԭ���ɶ�������������Ũ�ȵĽ��ͣ���NO���ɣ�ͼ2���м��װ�ÿɷ�ֹ�������������������������ķ�Ӧ��ʹ�õ�����������ȫ���գ�
��2��˫��ˮ�������ݲ�����˵�������������ڴ������·ֽ⣻
��3����ͭ��Ũ�����ڼ��������·�Ӧ��������ͭ�����������ˮ��
�ڼ��������أ����γ�������������0.1g��
�۸��ݱ������ݼ�������ͭ�ͽᾧˮ�����ʵ�������������xֵ��
��4��a��װ�����Ƿ�Һ©����
b�����������Һ�������ն�������
c����Һ���������SO2����Ʒ����ɫ������SO2����ɫ����������˵����NaHSO3��
d����Һ���������SO2����Ʒ����ɫ��SO2������NaOH��Ӧ����NaHSO3��
e���������ơ����������ơ����������Ժ�����ط�Ӧ��
��� �⣺��1���ɽ�����Ũ����ֶ�μӵ�ͭ����ϡ����Ļ�����п�֪����Һ©����ӦΪŨ���ᣬ
��װ�ñȽϿ�֪��װ��2�ж����װ����ʹ��Һ�뵼�ܲ�ֱ�ӽӴ�����ֹ������ͬʱNO���������Ӧ��ʹ�к������ܱ���ȫ���գ�
�ʴ�Ϊ��Ũ�����ֹ�������к������ܱ���ȫ���գ�
��2��˫��ˮ�������ݲ�����˵�������������ڴ������·ֽ⣻
�ʴ�Ϊ��ͭ���Ӷ�˫��ˮ�ֽ��д����ã�
��3����ͭ��Ũ�����ڼ��������·�Ӧ��������ͭ�����������ˮ����Ӧ�Ļ�ѧ����ʽΪCu+2H2SO4$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��
�ʴ�Ϊ��Cu+2H2SO4$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��
�ڸ���ʵ��������������ʼ���ʧˮ������������γ��������������0.1g��
�ʴ�Ϊ�����γ���������0.1g��
�۸�ʵ�龧�������Ϊ23.3g-11.7g=11.6g������ͭ������Ϊ19.7g-11.7g=8.0g��ˮ������Ϊ��11.6g-8.0g=3.6g����n��CuSO4��=$\frac{8.0g}{160g/mol}$=0.05mol��n��H2O��=$\frac{3.6g}{18g/mol}$=0.2mol������x=$\frac{0.2mol}{0.05mol}$=4��
����4��
��4��a����װ��ʹ�õIJ��������У��ƾ��ơ������ܡ���Һ©����Բ����ƿ����a����
b��KMnO4��Һ����β��������������������Ⱦ�����岻���ŷŵ������У����������Һ�������ն�������b��ȷ��
c������Ʒ����Һ���뵽��ƿ�У���Һ���������SO2��Ʒ����ɫ������SO2����ɫ����������˵����NaHSO��3����c����
d������Ʒ����Һ���뵽��ƿ�У���Ʒ����ɫ��˵����Һ�к��ж����������Ķ����������������ȫ����Ӧ��˵��NaOH����ȫת��ΪNaHSO3����d��ȷ��
e������Ʒ����Һ�������Ը��������Һ�������뵽��ƿ�У������Ϻ�ɫ��������ط�����Ӧ�������������ơ����������ơ����������Ժ�����ط�Ӧ������˵��NaOH����ȫת��ΪNaHSO3����e����
�ʴ�Ϊ��bd��
���� ���⿼��ͭ��Ũ����ķ�Ӧ���漰���ʵ���������������ʵ���������ѧ���㣬����ʵ�������ǽ���ؼ�����Ŀ�Ѷ��еȣ�

 �����ߴ���ϵ�д�
�����ߴ���ϵ�д�| A�� | 13.2mol•L-1 | B�� | 5.85mol•L-1 | C�� | 23.4mol•L-1 | D�� | 11.7mol•L-1 |
| A�� | �������е����Ų�ʽ��ԭ���У���1s22s22p63s23p2����1s22s22p3����1s22s22p2����1s22s22p63s23p1��ԭ�Ӱ뾶�ܣ��٣��ۣ��� | |
| B�� | �������м۵����Ų�ʽ��ԭ���У���1s22s22p63s2����1s22s22p63s23p1����1s22s2 2p3����1s22s22p4����һ�����ܣ��ۣ��ܣ��٣��� | |
| C�� | ��Na��K��Rb����N��P��As����O��S��Se ����Na��P��Cl��Ԫ�صĵ縺����ԭ������������������Ǣ� | |
| D�� | ijԪ����̬��̬ԭ�ӵ������ܣ�kJ•mol-1���ֱ�Ϊ738��1451��7733��10540��13630��17 995��21703��������������Ӧʱ�������ɵ���������X3+ |
| A�� | H2O��g���TH2��g��+$\frac{1}{2}$O2��g����H=+242 kJ•mol-1 | B�� | 2H2��g��+O2��g���T2H2O��l����H=-484 kJ•mol-1 | ||
| C�� | H2��g��+$\frac{1}{2}$O2��g���TH2O��g����H=+242 kJ•mol-1 | D�� | 2H2��g��+O2��g���T2H2O��g����H=+484 kJ•mol-1 |
| A�� | ����Cl2 | B�� | ����������Һ | C�� | ���� | D�� | ��Һ |