��Ŀ����
Ϊ̽��Fe(NO3)2���������ȷֽ����Ͳ�������ʣ�ij��ѧС�鿪չ����̽����
���������ϡ�2KNO3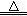 2KNO2+O2�� Fe(NO3)2
2KNO2+O2�� Fe(NO3)2 FexOy+NO2��+O2��
FexOy+NO2��+O2��
ʵ��һ��̽��Fe(NO3)2�ȷֽ�����������Ԫ�صļ�̬����С���ͬѧ���ֽ��Ĺ����������������ϡH2SO4�õ���Ӧ������Һ����������̽��ʵ�顣
��1�����ᴿ���롿
����һ����Ԫ��ֻ��+2�ۣ�
���������Ԫ�� ��
����������Ԫ�ؼ���+2������+3�ۡ�
��ʵ�����������һ����Һ�е���KSCN��Һ������һ����Һ�е�������KMnO4ϡ��Һ��
��2����ʵ������ʵ��� ��ʵ��� ��
��3����ʵ����ۡ��������������Fe(NO3)2�ֽ�Ļ�ѧ����ʽ�� ��
ʵ�����
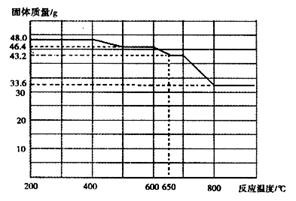
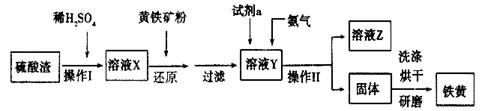
��4��̽��Fe(NO3)2�ȷֽ������������ʡ�С����ͬѧ����������ʵ�飬�����ʵ���ȱ�������ݡ���ѡ�Լ�����Ʒ��ŨH2SO4��Һ��4mol/LNaOH��Һ��0.1mol/LBaCl2��Һ�������ǵ�ľ����0.1mol/L����KMnO4��Һ������ˮ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ����Fe(NO3)2�������Թ��У����ȷֽ⡣ | ��˵���ֽ�����������к���NO2�� |
| ����2������������������ͨ��ʢ������ ��Ũ�����ϴ��ƿ�� �����һ�����ڼ��顣 | ��˵���ֽ�����������к�O2�� |
ʵ������KNO3�л���Fe(NO3)2��Ϊȷ��������Ԫ�صĺ�����С���ͬѧ��������ʵ�飺��ȡ�������Ʒ10g����ּ��ȷֽ⣻�ڽ���������ܽ⡢���ˣ�ȡ��������ϴ�ӡ�����Ƶ�������Ϊ3.2g������������Ԫ�ص���������Ϊ ����������λ��Ч���֣����ԭ��������Fe-56 O-16��
��17�֣���1��ֻ��+3�ۣ�1�֣�
��2����Һ����Ѫ��ɫ��2�֣�������������Ҳ���֣� ��Һ�Ϻ�ɫ����ɫ��2�֣�
��3��4Fe(NO3)2 2Fe2O3+8NO2��+O2����2�֣���ѧʽ�������֣�ϵ������1�֣���д������1�֣�
2Fe2O3+8NO2��+O2����2�֣���ѧʽ�������֣�ϵ������1�֣���д������1�֣�
��4��ʵ�鲽�� Ԥ������ͽ��� ����1��ȡ����Fe(NO3)2�������Թ��У����ȷֽ⡣ �к���ɫ���������2�֣�������������Ҳ���֣���˵���ֽ�����������к���NO2�� ����2������������������ͨ��ʢ������ 4mol/LNaOH��Һ��2�֣���Ũ�����ϴ��ƿ���ô����ǵ�ľ����2�֣������һ�����ڼ��顣 �����ǵ�ľ����ȼ��1�֣���˵���ֽ�����������к�O2��
��5��22.4%
���������������1������������֪�IJ����ƶϣ��������Ϊ��Ԫ��ֻ��+3�ۣ���2������ʵ������ƶϣ�Fe(NO3)2���ȷֽ�����Ĺ������ֻ��Fe2O3��Fe2O3������ϡ���ᷴӦ����Fe3+����Һ����Fe3+��KSCN��Һ��죬����ʹ���Ը��������Һ��ɫ����Fe2O3��ϵ��Ϊ1����������������ԭ�Ӹ����غ���ƽ���÷�ӦΪ2Fe(NO3)2 Fe2O3+4NO2��+1/2O2����ϵ���ӱ��ɵã�4Fe(NO3)2
Fe2O3+4NO2��+1/2O2����ϵ���ӱ��ɵã�4Fe(NO3)2 2Fe2O3+8NO2��+O2������4��Fe(NO3)2���ȷֽ��������������У�NO2�Ǻ���ɫ���壬O2����ɫ��ζ���壬�к���ɫ�������������˵���ֽ�����������к���NO2������NO2�ж���������ˮ��Ӧ��������O2��Ӧ��NO����˲���2��Ӧ�����ù���4mol/LNaOH��Һ���ն����NO2������Ũ�����������һ���������ô����ǵ�ľ�����飬��������ľ����ȼ��˵���ֽ�����������к���O2����5�����������֪��������m(Fe2O3)=3.2g��������Ԫ���غ�ɵù�ϵʽ��2Fe��Fe2O3����ԭ����m(Fe)= m(Fe2O3)��2��56/160=3.2g��2��56/160�����ڻ����������Ϊ10g������������Ԫ�ص���������Ϊ3.2g��2��56/160��10g��100%=22.4%��
2Fe2O3+8NO2��+O2������4��Fe(NO3)2���ȷֽ��������������У�NO2�Ǻ���ɫ���壬O2����ɫ��ζ���壬�к���ɫ�������������˵���ֽ�����������к���NO2������NO2�ж���������ˮ��Ӧ��������O2��Ӧ��NO����˲���2��Ӧ�����ù���4mol/LNaOH��Һ���ն����NO2������Ũ�����������һ���������ô����ǵ�ľ�����飬��������ľ����ȼ��˵���ֽ�����������к���O2����5�����������֪��������m(Fe2O3)=3.2g��������Ԫ���غ�ɵù�ϵʽ��2Fe��Fe2O3����ԭ����m(Fe)= m(Fe2O3)��2��56/160=3.2g��2��56/160�����ڻ����������Ϊ10g������������Ԫ�ص���������Ϊ3.2g��2��56/160��10g��100%=22.4%��
���㣺����̽��ʵ�鷽������ƺͶ���ʵ��ļ��㣬�漰������롢��֤���롢���ʵ�鷽������ƽ��ѧ����ʽ�����������Ԫ�����������IJⶨ��֪ʶ��

 ��Ч���ܿ�ʱ��ҵϵ�д�
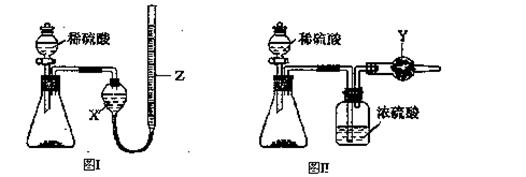
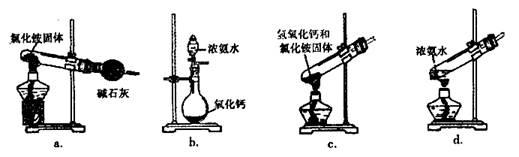
��Ч���ܿ�ʱ��ҵϵ�д�ij̽��С�������ͼ��ʾװ�ý���Fe����ˮ�����ķ�Ӧ��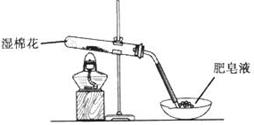
��1��ʵ��ǰ���װ�������Եķ���Ϊ________________________________________________________��
��2������ʵ�������������ʵ�������_____________________________________________��
��3����̽��С���Ϊ���飬����ͼװ�ý��жԱ�ʵ�飬�����þƾ���ơ������þƾ��Ƽ��ȣ���Ӧ�����Ϊ��ɫ��ĩ(������)������ֱ��ò����������ʵ�顣
| ���� | ���� | �������� | �������� |
| 1 | ȡ��ɫ��ĩ����ϡ���� | �ܽ⣬������ | �ܽ⣬������ |
| 2 | ȡ����1����Һ���μ�����KMnO4��Һ | ��ɫ��ȥ | ��ɫ��ȥ |
| 3 | ȡ����1����Һ���μ�KSCN��Һ | ��� | ������ |
| 4 | ����3��Һ�еμ�������ˮ | ��ɫ��ȥ | �ȱ�죬����ɫ |
������õ��ĺ�ɫ��ĩ�� ��
�ڼ��鲽��1�з�Ӧ�����ӷ���ʽΪ ��
�����鲽��4�У���Һ����ԭ��Ϊ ����Һ��ɫ���ܵ�ԭ���� ����֤����Ϊ ��
�Ȼ�������Һ�еμ����軯����Һ���ٵμ�˫��ˮ����������Ѫ��ɫ�����ɫ����ȥ�ʻ�ɫ���������ݲ�������Ը�ʵ������ijʵ��С���ͬѧ������̽��
һ���������
����A��������H2O2�ֽ������O2
����B��������KSCN������ΪN2��SO2��CO2��
����C����ɫ��ȥ��ԭ����KSCN������������������
����ʵ��̽��
̽��1
| ʵ����� | ʵ������ | ���� |
| ��1mL 0.1mol/L��FeCl2��Һ�м�2��KSCN��Һ | ��Һ����� | Fe2+��SCN-����� |
| ����ٵ���Һ�м�3%��H2O21�β��� | ��������Ѫ��ɫ ����ɫ | ����H2O2����Һ�� ������ (���ӷ���) |
| ����ڵ���Һ�м�����H2O2��Һ | ��Һ�г��ִ������� Ѫ��ɫ��ȥ | |
| ���ô����ǵ�ľ��������е����� | ľ����ȼ | ����� ���� |
̽��2
| ʵ����� | ʵ������ | ���� |
| ��ȡ2ml KSCN��Һ�����м��뼸��BaCl2��Һ��ϡ���� | ���������� | |
| ��������õ���Һ�еμ�3%��H2O2 | ��Һ�г��ְ�ɫ���������������� | ��ɫ����ΪBaSO4 |
| �۽�6%��H2O2��Һ����KSCN�����У����ɵ���������ͨ��Ʒ����Һ������KMnO4��Һ�ͳ����ʯ��ˮ | | KSCN��H2O2����������SO2��CO2���� |
(1)̽��1�У�H2O2�ֽ��ٶȺܿ��ԭ��
(2)̽��1�У�˵����ԭ��Fe2+ SCN-
(3)̽��2�У�����KMnO4��Һ��������
��֤��������CO2��������
(4)��SCN -��H2O2�����õ�N2��SO2��CO2��SO42-����SO2��SO42-�����ʵ�����Ϊ1:1����д���÷�Ӧ�����ӷ���ʽ
�������岻����ŨH2SO4�������
| A��CO2 | B��SO2 | C��NH3 | D��HC1 |
úȼ�յ������к���CO��SO2���壬���й��������������˵����ȷ����
| A�����߶�������ˮ |
| B�����߶����ж����� |
| C�����߶���ʹƷ����Һ��ɫ |
| D�����߶����γ��������Ҫԭ�� |
 �ķ����� ��
�ķ����� ��