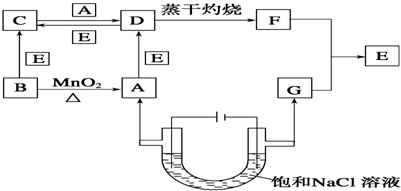
ЬтФПФкШн
NAЮЊАЂЗќМгЕТТоГЃЪ§ЕФЪ§жЕЃЌЯТСаЫЕЗЈжае§ШЗЕФЪЧЃЈЁЁЁЁЃЉ
| AЁЂдкУмБеШнЦїжаМгШы1.5mol H2КЭ0.5mol N2ЃЌГфЗжЗДгІКѓПЩЕУЕНNH3ЗжзгЪ§ЮЊNA |
| BЁЂвЛЖЈЬѕМўЯТЃЌ2.3gЕФNaЭъШЋгыO2ЗДгІЩњГЩ3.6gВњЮяЪБЕУЕНЕФЕчзгЪ§ЮЊ0.1NA |
| CЁЂ1.0LЕФ0.1mol?L-1 Na2SШмвКжаКЌгаЕФNa+РызгЪ§ЮЊ0.2NA |
| DЁЂБъзМзДПіЯТЃЌ22.4LЕФCCl4жаКЌгаЕФCCl4ЗжзгЪ§ЮЊNA |
ПМЕуЃКАЂЗќМгЕТТоГЃЪ§
зЈЬтЃКАЂЗќМгЕТТоГЃЪ§КЭАЂЗќМгЕТТоЖЈТЩ
ЗжЮіЃКAЁЂЕЊЦјКЭЧтЦјЕФЗДгІЪЧПЩФцЗДгІВЛФмНјааГЙЕзЃЛ
BЁЂвРОнЕчзгЪиКуЃЌНсКЯЭъШЋЗДгІЕФФЦМЦЫуЕчзгзЊвЦЃЛ
CЁЂвРОнn=CVМЦЫуЮяжЪЕФСПМЦЫуШмжЪжаЫљКЌРызгЪ§ЃЛ
DЁЂБъзМзДПіCCl4ВЛЪЧЦјЬхЃЎ
BЁЂвРОнЕчзгЪиКуЃЌНсКЯЭъШЋЗДгІЕФФЦМЦЫуЕчзгзЊвЦЃЛ
CЁЂвРОнn=CVМЦЫуЮяжЪЕФСПМЦЫуШмжЪжаЫљКЌРызгЪ§ЃЛ
DЁЂБъзМзДПіCCl4ВЛЪЧЦјЬхЃЎ
НтД№ЃК
НтЃКAЁЂЕЊЦјКЭЧтЦјЕФЗДгІЪЧПЩФцЗДгІВЛФмНјааГЙЕзЃЌдкУмБеШнЦїжаМгШы1.5mol H2КЭ0.5mol N2ЃЌГфЗжЗДгІКѓПЩЕУЕНNH3ЗжзгЪ§аЁгкNAЃЌЙЪAДэЮѓЃЛ
BЁЂвРОнЕчзгЪиКуЃЌНсКЯЭъШЋЗДгІЕФФЦМЦЫуЕчзгзЊвЦЃЌ2.3gЕФNaЮяжЪЕФСПЮЊ0.1molЃЌЭъШЋгыO2ЗДгІЃЌЪЇЕчзг0.1molЃЌЩњГЩ3.6gВњЮяЪБЪЇШЅЕФЕчзгЪ§ЮЊ0.1NAЃЌЙЪBДэЮѓЃЛ
CЁЂМЦЫуШмжЪЮяжЪЕФСПn=CV=1.0LЁС0.1mol/L=0.1molЃЌNa2SШмвКжаКЌгаЕФNa+РызгЪ§ЮЊ0.2NAЃЌЙЪCе§ШЗЃЛ
DЁЂБъзМзДПіCCl4ВЛЪЧЦјЬхЃЌ22.4LЕФCCl4ЮяжЪЕФСПВЛЪЧ1molЃЌЙЪDДэЮѓЃЛ
ЙЪбЁCЃЎ
BЁЂвРОнЕчзгЪиКуЃЌНсКЯЭъШЋЗДгІЕФФЦМЦЫуЕчзгзЊвЦЃЌ2.3gЕФNaЮяжЪЕФСПЮЊ0.1molЃЌЭъШЋгыO2ЗДгІЃЌЪЇЕчзг0.1molЃЌЩњГЩ3.6gВњЮяЪБЪЇШЅЕФЕчзгЪ§ЮЊ0.1NAЃЌЙЪBДэЮѓЃЛ
CЁЂМЦЫуШмжЪЮяжЪЕФСПn=CV=1.0LЁС0.1mol/L=0.1molЃЌNa2SШмвКжаКЌгаЕФNa+РызгЪ§ЮЊ0.2NAЃЌЙЪCе§ШЗЃЛ
DЁЂБъзМзДПіCCl4ВЛЪЧЦјЬхЃЌ22.4LЕФCCl4ЮяжЪЕФСПВЛЪЧ1molЃЌЙЪDДэЮѓЃЛ
ЙЪбЁCЃЎ
ЕуЦРЃКБОЬтПМВщСЫПЩФцЗДгІЗжЮіЃЌбѕЛЏЛЙдЗДгІЕчзгзЊвЦМЦЫугІгУЃЌЦјЬхФІЖћЬхЛ§ЬѕМўгІгУЃЌеЦЮеЛљДЁЪЧЙиМќЃЌЬтФПНЯМђЕЅЃЎ

СЗЯАВсЯЕСаД№АИ
ЯрЙиЬтФП
дкКЌгаFeCl3КЭBaCl2ЕФЫсадШмвКжаЃЌЭЈШызуСПЕФSO2КѓгаАзЩЋГСЕэЩњГЩЃЌЙ§ТЫКѓЃЌЯђШмвКжаЕЮМгKSCNШмвКЃЌЮоУїЯдЯжЯѓЃЌЯТСаа№Ъіе§ШЗЕФЪЧЃЈЁЁЁЁЃЉ
| AЁЂFe3+ШЋВПБЛЛЙдЮЊFe2+ |
| BЁЂШмвКЕФЫсадМѕШѕ |
| CЁЂFe3+ВПЗжБЛЛЙдЮЊFe2+ |
| DЁЂАзЩЋГСЕэЪЧBaSO4КЭBaSO3 |
ЯТСаРызгЗНГЬЪНФмЙЛе§ШЗБэЪОЫЎНтЗДгІЕФЪЧЃЈЁЁЁЁЃЉ
| AЁЂHCO3-+H2O?CO32-+H3O+ |
| BЁЂCO32-+H2O?HCO3-+2OH- |
| CЁЂCu2++2H2O?CuЃЈOHЃЉ2+2H+ |
| DЁЂCH3COO-+H+=CH3COOH |
дкЫЎШмвКжаФмДѓСПЙВДцЕФРызгзщЪЧЃЈЁЁЁЁЃЉ
| AЁЂAl3+ЁЂH+ЁЂHCO3- |
| BЁЂNa+ЁЂNO3-ЁЂCl- |
| CЁЂH+ЁЂOH-ЁЂSO42- |
| DЁЂCa2+ЁЂK+ЁЂCO32- |
ЯТСаИїзщРызгдкжИЖЈШмвКжаФмДѓСПЙВДцЕФЪЧЃЈЁЁЁЁЃЉ
| AЁЂМгШыЯЁЯѕЫсЕФШмвКЃКK+ЁЂCl-ЁЂHS-ЁЂSO32- |
| BЁЂМгШыFeCl3ШмвКЃКI-ЁЂNH4+ЁЂClЁЂBa2+ |
| CЁЂМгШыNaOHЕФШмвКЃКNa+ЁЂAlO2-ЁЂCO32-ЁЂK+ |
| DЁЂЮоЩЋpH=12ЕФШмвКЃКFe3+ЁЂMg2+ЁЂAl3+ЁЂNO3- |
ЭЌЮТЭЌбЙЯТЃЌЯрЭЌЮяжЪЕФСПЕФCOКЭN2ЃЌОпгаЯрЭЌЕФЃЈЁЁЁЁЃЉ
| AЁЂдзгЪ§ | BЁЂжЪзгЪ§ |
| CЁЂЬхЛ§ | DЁЂжЪСП |
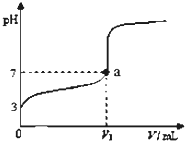 ГЃЮТЯТЃЌЯђ20mL x mol?L-1 CH3COOHШмвКжаж№ЕЮМгШыЕШЮяжЪЕФСПХЈЖШЕФ
ГЃЮТЯТЃЌЯђ20mL x mol?L-1 CH3COOHШмвКжаж№ЕЮМгШыЕШЮяжЪЕФСПХЈЖШЕФNaOHШмвКЃЌЛьКЯвКЕФpHЫцNaOHШмвКЕФЬхЛ§ЃЈVЃЉБфЛЏЙиЯЕШчЭМЫљЪОЃЈКіТдЮТЖШБфЛЏЃЉЃЎЯТСаЫЕЗЈжаВЛе§ШЗЕФЪЧЃЈЁЁЁЁЃЉ
| AЁЂЩЯЪі CH3COOHШмвКжаЃКcЃЈH+ЃЉ=1ЁС10-3mol?L-1 |
| BЁЂЭМжаV1ЃО20mL |
| CЁЂaЕуЖдгІЕФШмвКжаЃКcЃЈCH3COO-ЃЉ=cЃЈNa+ЃЉ |
| DЁЂМгШыNaOHШмвКЬхЛ§ЮЊ20 mLЪБЃЌШмвКжаЃКcЃЈCH3COOHЃЉ+cЃЈH+ЃЉ=cЃЈOH-ЃЉ+cЃЈCH3COO-ЃЉ |