��Ŀ����
����˵������ȷ���ǣ�������
| A��ʵ�������Ҵ���Ũ���ᰴ�����3��1��ϼ��ȵ�170������ϩ |
| B��������ɱ����ֹʹ���ã���Щҩ����Ҳ�����������ı��ӣ��������ǿ��Խ��������ڻ���������ֱ��ͿĨ��Ƥ�������������� |
| C����ҵ�ƾ����ܼ�ˮ��������Ʒ���������к��м״�������Ҳ���о�����ζ�������������˵�˫Ŀ����ʧ�������� |
| D��ͨ����������һ�����ҵ�ʯ�ͻ�ѧ��ҵ��չˮƽ�ı�־�����͵IJ��� |
���㣺��ϩ��ʵ�����Ʒ�,��ϩ����;,���ӵĻ�ѧ����
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
������A��ʵ��������ˮ�ƾ���Ũ������1��3������Ȼ�ϼ�������ϩ���壻
B�������ж�����ɱ����������Ҳ��ʹ���嵰���ʱ��ԣ�
C����ҵ�ƾ��г����м״����״��о綾����ʳ�ã�
D��������ϩ����;��֪����ϩ���������������ϡ��ϳ���ά���л��ܼ��ȣ�����Ҫ�Ļ���ԭ�ϣ�
B�������ж�����ɱ����������Ҳ��ʹ���嵰���ʱ��ԣ�
C����ҵ�ƾ��г����м״����״��о綾����ʳ�ã�
D��������ϩ����;��֪����ϩ���������������ϡ��ϳ���ά���л��ܼ��ȣ�����Ҫ�Ļ���ԭ�ϣ�
���
�⣺A��ʵ��������ϩ���壬Ũ���������ˮ�ԣ��ٽ���Ӧ������Ӧ�����ƶ���Ũ���ỹ���������ӿ췴Ӧ���ʣ�������ˮ�ƾ���Ũ������1��3������ȣ���A����
B�������ж�����ʹ�����ʱ��Զ���ɱ���������������Խ�����ֱ��ͿĨ��Ƥ�������������ã�������˺����壬��B����
C���״��о綾��ʹ���ж������ԡ��۾�ʧ�����������������Թ�ҵ�ƾ��к��м״������ܹ����ƾƣ���C��ȷ��
D����ϩ���������������ϡ��ϳ���ά���л��ܼ��ȣ�����Ҫ�Ļ���ԭ�ϣ���ϩ��ҵ�ķ�չ��������������ʯ��Ϊԭ�ϵ�ʯ�ͻ����ķ�չ�����һ��������ϩ��ҵ�ķ�չˮƽ���ѳ�Ϊ�����������ʯ�ͻ�ѧ��ҵˮƽ����Ҫ��־����D����
��ѡC��
B�������ж�����ʹ�����ʱ��Զ���ɱ���������������Խ�����ֱ��ͿĨ��Ƥ�������������ã�������˺����壬��B����
C���״��о綾��ʹ���ж������ԡ��۾�ʧ�����������������Թ�ҵ�ƾ��к��м״������ܹ����ƾƣ���C��ȷ��
D����ϩ���������������ϡ��ϳ���ά���л��ܼ��ȣ�����Ҫ�Ļ���ԭ�ϣ���ϩ��ҵ�ķ�չ��������������ʯ��Ϊԭ�ϵ�ʯ�ͻ����ķ�չ�����һ��������ϩ��ҵ�ķ�չˮƽ���ѳ�Ϊ�����������ʯ�ͻ�ѧ��ҵˮƽ����Ҫ��־����D����
��ѡC��
���������⿼����ʵ��������ϩ�����ӡ���ϩ����;����ҵ�ƾ���ʹ�ã���Ŀ�ѶȲ���ע�������е��л��������Լ�֪ʶӦ�ã�ע�����֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 Сѧ��10���ӿ������100��ϵ�д�
Сѧ��10���ӿ������100��ϵ�д�
�����Ŀ
���з�Ӧ�����ӷ���ʽ��д��ȷ���ǣ�������
A����������Һ������ȩ�е�ȩ����CH3CHO+2Ag��NH3��2++2OH-
| ||
| B���ö�������ˮ��Һ������������SO2+Br2+2H2O=2H++SO42-+2HBr | ||
| C���ڣ�NH4��2SO4��Һ����μ���������Ba��OH��2��Һ��NH4++SO42-+Ba2++OH-=BaSO4��+NH3?H2O | ||
| D�����������Һ��ͨ������SO2���壺Ba2++2ClO-+SO2+H2O=CaSO3��+2HClO |
�������ʵĻ�ѧ���ﲻ��ȷ���ǣ�������
| A���������������ʽ��C2H4O |
B����ϩ�ı���ģ�ͣ� |
C��������ӵ����ģ�ͣ�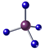 |
| D������Ľṹʽ��CH3CH3 |
��֪��Ӧ��2M��g��+N��g��+P��g��
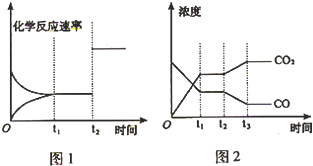
2Q��g��+3R��g����H��0���ں��ݵ��ܱ������У������й�˵����ȷ���ǣ�������
| 180�� |
| ���� |
| A��ƽ��ʱ�������������䣬����M��Ũ�ȣ�N��ת���ʼ�С |
| B��ƽ��ʱ�������������䣬�����¶ȿ�ʹ�÷�Ӧ��ƽ�ⳣ������ |
| C����λʱ��������N��Q�����ʵ�����Ϊ1��1ʱ����Ӧ�ﵽƽ�� |
| D�������������䣬ʹ�ø�Ч������N��P��ת�������� |
���������У������ڵ���ʵ��ǣ�������
| A��H2SO4 |
| B��Na2SO4 |
| C���Ҵ� |
| D��Ba��OH��2 |
NAΪ�����ӵ���������������������ǣ�������
| A��0.1mol Na2O2������ˮ��ȫ��Ӧʱ��ת�Ƶ�����ĿΪ0.1NA |
| B��12g���ʯ���еĹ��ۼ���Ϊ2NA |
| C��0.1mol��ͭ��������Ũ���ᷴӦ����SO2������������ԼΪ2.24L |
| D����1L 0.1mol?L-1̼������Һ������������С��0.1NA |
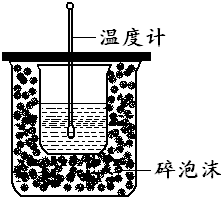 ����ͼ��ʾ��װ���н����кͷ�Ӧ�ķ�Ӧ�Ȳⶨ��ʵ��������£�
����ͼ��ʾ��װ���н����кͷ�Ӧ�ķ�Ӧ�Ȳⶨ��ʵ��������£�