��Ŀ����
�Իش��������⣺
��1����֪24��A��40��Bǡ����ȫ��Ӧ����0.8molC��32��D����C��Ħ������Ϊ ��
��2��ͼ1Ϊʵ����ijŨ�����Լ�ƿ�ı�ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺

�ٸ�Ũ������HCl�����ʵ���Ũ��Ϊ mol/L��
����ʵ��������450mL 2.38mol/L��ϡ���ᣬ���ø�Ũ���� mL����ͼ2��ʾ��������������Һ�϶�����Ҫ���� ������ţ�������������Һ�����õ��IJ��������� �����������ƣ�����ʵ����������������ȷ��������ʱ���ӿ̶��ߣ���������ҺŨ�� 2.38mol/L ������ڡ������ڡ���С�ڡ�����ͬ����
��1����֪24��A��40��Bǡ����ȫ��Ӧ����0.8molC��32��D����C��Ħ������Ϊ
��2��ͼ1Ϊʵ����ijŨ�����Լ�ƿ�ı�ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺

�ٸ�Ũ������HCl�����ʵ���Ũ��Ϊ
����ʵ��������450mL 2.38mol/L��ϡ���ᣬ���ø�Ũ����
���㣺��Һ������
ר�⣺ʵ����
��������1�����������غ㶨�ɼ����C��������Ȼ�����M=
�����C��Ħ��������
��2���ٸ���c=
�������Ũ��������ʵ���Ũ�ȣ�
������450mLϡ���ᣬС��ѡ��500mL����ƿ��ʵ�������Ƶ���500mL 2.38mol/L��ϡ���ᣬ����ϡ���������ʵ����ʵ�������������ҪŨ������������������һ�����ʵ���Ũ�ȵ���Һ����ѡ��������Ȼ���ж���Ҫ����������ȱ�ٵ����������Ӷ��ݣ����������ˮ����������ƿ�Ŀ̶��ߣ�
| m |
| n |
��2���ٸ���c=
| 1000��w |
| M |
������450mLϡ���ᣬС��ѡ��500mL����ƿ��ʵ�������Ƶ���500mL 2.38mol/L��ϡ���ᣬ����ϡ���������ʵ����ʵ�������������ҪŨ������������������һ�����ʵ���Ũ�ȵ���Һ����ѡ��������Ȼ���ж���Ҫ����������ȱ�ٵ����������Ӷ��ݣ����������ˮ����������ƿ�Ŀ̶��ߣ�
���
�⣺��1��24��A��40��Bǡ����ȫ��Ӧ����0.8molC��32��D�����������غ��֪0.8molC������Ϊ��40g+24g-32g=32g����C��Ħ������Ϊ��
=40g/mol��
�ʴ�Ϊ��40g/mol��
��2���ٸ�Ũ������HCl�����ʵ���Ũ��Ϊ��
mol/L=11.9mol/L��
�ʴ�Ϊ��11.9��
����ʵ��������450mL 2.38mol/L��ϡ���ᣬʵ����û�й��Ϊ450mL������ƿ����Ҫѡ��500mL������ƿ������500mL 2.38mol/L��ϡ���ᣬ��Ҫ��Ũ��������Ϊ��
=0.1L=100mL��
����500mL 2.38mol/L��ϡ���ᣬ��Ҫʹ�õ������У���Ͳ���ձ�����������500mL����ƿ����ͷ�ιܣ�����Ҫ������Ϊ��AԲ����ƿ��C��Һ©������ȱ���ձ��Ͳ�������
������Һ�����У������������ƿ�Ŀ̶��߶��ݣ��ᵼ�¼��������ˮ���ƫ�����Ƶ���Һ���ƫ����ҺŨ��ƫ�ͣ���С��2.38mol/L��
�ʴ�Ϊ��100�� A��C���ձ�����������С�ڣ�
| 32g |
| 0.8mol |
�ʴ�Ϊ��40g/mol��
��2���ٸ�Ũ������HCl�����ʵ���Ũ��Ϊ��
| 1000��1.19��36.5% |
| 36.5 |
�ʴ�Ϊ��11.9��
����ʵ��������450mL 2.38mol/L��ϡ���ᣬʵ����û�й��Ϊ450mL������ƿ����Ҫѡ��500mL������ƿ������500mL 2.38mol/L��ϡ���ᣬ��Ҫ��Ũ��������Ϊ��
| 2.38mol/L��0.5L |
| 11.9mol/L |
����500mL 2.38mol/L��ϡ���ᣬ��Ҫʹ�õ������У���Ͳ���ձ�����������500mL����ƿ����ͷ�ιܣ�����Ҫ������Ϊ��AԲ����ƿ��C��Һ©������ȱ���ձ��Ͳ�������
������Һ�����У������������ƿ�Ŀ̶��߶��ݣ��ᵼ�¼��������ˮ���ƫ�����Ƶ���Һ���ƫ����ҺŨ��ƫ�ͣ���С��2.38mol/L��
�ʴ�Ϊ��100�� A��C���ձ�����������С�ڣ�
���������⿼��������һ�����ʵ���Ũ�ȵ���Һ�ķ�����Ϊ�е��Ѷȵ����⣬ע��������������һ�����ʵ���Ũ�ȵ���Һ���������������ǿ��ע������ԣ����ض�ѧ�������������ͽ��ⷽ����ָ����ѵ��������������ѧ������˼ά�������Ͻ��Ĺ淶ʵ�����������

��ϰ��ϵ�д�
 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д� �ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�����Ŀ
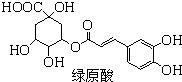 ��ԭ����һ���н�ѹ���õ�ҩ����ɿ����ᣨһ�ַ����ᣩ�������ͨ��������Ӧ�ϳɣ�����˵����ȷ���ǣ�������
��ԭ����һ���н�ѹ���õ�ҩ����ɿ����ᣨһ�ַ����ᣩ�������ͨ��������Ӧ�ϳɣ�����˵����ȷ���ǣ�������| A�����������ʽΪC9H8O4 |
| B��1mol��������NaOH��Һ��Ӧ���������5mol NaOH��1mol��ԭ����Br2ˮ��Ӧ���������6mol Br2 |
| C����ԭ���ܷ���ȡ�����ӳɡ���ȥ�ͻ�ԭ��Ӧ |
| D��������Ϳ����ᶼ����FeCl3��Һ������ɫ��Ӧ |
����˵������ȷ���ǣ�������
| A�������£���ˮ����ε�����������Һ��pH=7������Һ�У�c��NH4+��=c��Cl-�� |
| B���������pH=4������ʹ���ϡ�ͳ�pH=5����Һ��������������ˮ���� |
| C��pH=13��NaOH��Һ��pH=1�Ĵ�����Һ�������Ϻ�������Һ��pH��7 |
| D��0.2mol?L-1��NaHCO3��Һ�У�c��Na+����c��HCO3-����c��OH-����c��H+�� |
ij����һ��ͬ���칹�壬�˴Ź������ײ�÷�����ֻ��һ�����շ壬������ķ���ʽ����Ϊ��������
| A��C15H32 |
| B��C16H34 |
| C��C17H36 |
| D��C18H38 |
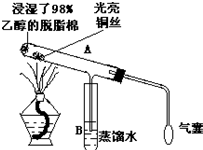 �йش����Ĵ�����������ɴӡ��Ҵ���������ʵ��õ�һЩ��ʶ����ʵ��װ����ͼ��
�йش����Ĵ�����������ɴӡ��Ҵ���������ʵ��õ�һЩ��ʶ����ʵ��װ����ͼ�� A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��������������ֲ�ͬ����ش�
A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��������������ֲ�ͬ����ش�