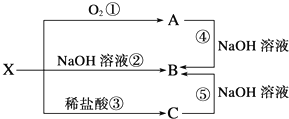
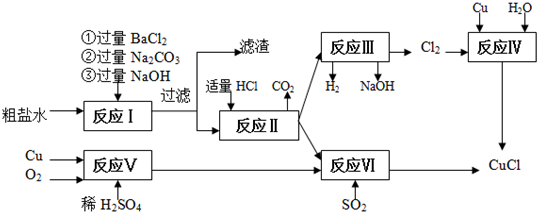
��Ŀ����
6�������뻯ѧ�йص�˵���д�����ǣ�������| A�� | �����Ƽ��������ȡ���չ����о�Ӧ���������ܽ�ȵIJ��� | |
| B�� | ʹ�ÿ�������Դ���ó��ٽ������̼����л��ܼ���ע��ԭ�ӵľ����ԡ����õ��ܺ��������յȶ�����ɫ��ѧ������ | |
| C�� | ʯ�ͷ���ú����������ˮɹ�Ρ���ȥ���ۡ���������ȡ�����͵ȹ��̶��������仯 | |
| D�� | �Ͻ�������ָ�ۺ����ֻ��������ϵĽ���Ԫ�ز��γɾ��г������Եij������ϣ�����һ�ѣ�Nb-Ti���Ͻ������۵�һ��ȴ���Ҫ�� |
���� A�������Ƽ�ǽ�CO2��NH3ͨ�뱥��NaCl��Һ�У�����NaHCO3�ܽ��С��Na2CO3����NaHCO3����Һ��������
B����ɫ��ѧ���Ի����Ѻ��ͻ�ѧ��Ҫ��ԭ�����������е�ԭ����ȫ��������ȫ��ת�������IJ�Ʒ�У�ԭ�ӵ�������100%��
C��ú�������ǽ���ת��Ϊ��ȼ����Ĺ��̣���Ҫ��ӦΪ̼��ˮ������Ӧ����H2��CO������Ĺ��̣������������ɣ����ڻ�ѧ�仯��
D���Ͻ���۵�һ���������Ľ����ĵͣ�
��� �⣺A�������Ƽ�ǽ�CO2��NH3ͨ�뱥��NaCl��Һ�У��������·�Ӧ��NH3+CO2+H2O=NH4HCO3��NH4HCO3+NaCl=NH4Cl+NaHCO3��������NaHCO3�ܽ����С������NaHCO3�ľ�����������A��ȷ��
B��ʹ�ÿ�������Դ���ó��ٽ������̼����л��ܼ���ע��ԭ�ӵľ����ԡ����õ��ܺ��������յȷ�����ɫ��ѧ��Ҫ��B��ȷ��
C��ʯ�ͷ���ˮɹ�Ρ���������ȡ�����͵ȹ��̶��������仯����ú���������������ʵ����ɣ����ڻ�ѧ�仯����C����
D���Ͻ���۵�һ���������Ľ����ĵͣ���һ�ѣ�Nb-Ti���Ͻ������۵�һ��ȴ���Ҫ����ȷ����D��ȷ��
��ѡC��
���� �����漰�����Ƽ��ɫ��ѧ��ú�������Լ��Ͻ��֪ʶ��ע��֪ʶ�Ĺ��ɺ������ǹؼ����ѶȲ���

 ����С��ʿ���������ϵ�д�
����С��ʿ���������ϵ�д�| A�� | ����ϡ���ᷴӦ��Fe+2H+�TFe3++H2�� | |
| B�� | ���������������Һ��Ӧ��Ba2++SO42-�TBaSO4�� | |
| C�� | ʳ��ˮ�еμ���������Һ��Ag++Cl-�TAgCl�� | |
| D�� | ʯ��ʯ�����ᷴӦ��CO32-+2H+�TCO2��+H2O |
| A�� | ̼��������Һ�У�K+��NO3-��Cl-��H+ | |
| B�� | ʹ��̪��Һ�ʺ�ɫ����Һ�У�Mg2+��Cu2+��SO42-��K+ | |
| C�� | ����������Һ�У�Cu2+��Mg2+��SO42-��Cl-- | |
| D�� | ʹʯ����Һ������Һ�У�Na+��Cl-��K+��CH3COO- |
| A�� | c��H+��=0.1mol/L����Һ��Na+��NH4+��SO42-��S2O32- | |
| B�� | $\frac{c��{H}^{+}��}{c��O{H}^{-}��}$=1��10-12����Һ��K+��AlO2-��CO32-��Na+ | |
| C�� | c��Fe2+��=0.1mol/L����Һ��H+��Al3+��NO3-��SCN- | |
| D�� | ����ˮ�������c��H+��=1��10-12mol/L����Һ��Fe3+��ClO-��Na+��SO42- |
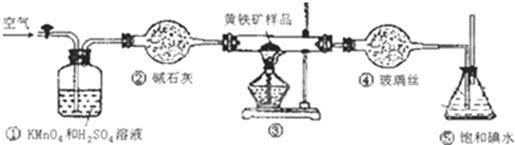
ʵ�����ȷ�����������£�
A�����Ӻ�װ�ã������װ�õ�������
B����ȡ��ϸ�Ļ�������Ʒ
C����2.0g��ƷС�ĵط���Ӳ�ʲ�������
D����1L/min�����ʹ������
E����Ӳ�ʲ������еĻ���������Ʒ���ȵ�800�桫850��
F����300mL�ı��͵�ˮ����SO2�������ķ�Ӧ�ǣ�I2+SO2+2H2O=2HI+H2SO4
G������Һ��CCl4��ȡ
H��ȡ20.00mL����Һ���ñ�0.2000mol•L-1��NaOH��Һ�ζ���
�Իش�
��1��д��װ�â��з�����Ӧ�Ļ�ѧ����ʽ4FeS2+11O2$\frac{\underline{\;\;��\;\;}}{\;}$2Fe2O3+8SO2��
��2������G��������Ҫ�����Ƿ�Һ©����Ӧȡ�ϣ���ϡ����¡�������Һ���к���ʵ�飮
��3��װ�âٵ������dz�ȥ�����еĻ�ԭ�����壬���ʵ���ȷ�ʣ�
��4������H�еζ�ʱӦѡ�÷�̪��ָʾ�������Ը��ݵ������һ��NaOH��Һ����Һ��Ϊdz��ɫ�����ڰ�����ڲ���ɫ�������жϵζ��Ѿ��ﵽ�յ㣮
��5���ٶ����������е����ڲ���E����ȫ��ת��ΪSO2�����ұ����͵�ˮ��ȫ���գ��ζ��õ������������ʾ��
| �ζ����� | ����Һ������/mL | ������Һ�����/mL | |
| �ζ�ǰ | �ζ��� | ||
| ��һ�� | 20.00mL | 0.00 | 20.98 |
| �ڶ��� | 20.00mL | 0.22 | 20.20 |
| ������ | 20.00mL | 0.36 | 20.38 |
��6��Ҳ��������á��������������ⶨ�������к��������������������ַ����ⶨ���������װ�â���������Һ�м������������Լ�B��
A����������Һ B���Ȼ�����Һ C������ʯ��ˮ D�����Ը��������Һ��