��Ŀ����
����Ŀ������һ�ֺܻ��õ�Ԫ���������˵����������γ�2��+6��+4��+2��+1�۵Ļ����
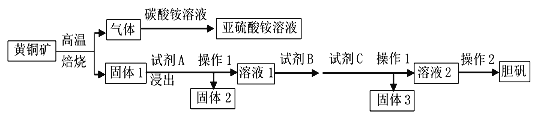
��.����������(Na2S2O5)�dz��õ�ʳƷ��������֮һ������ǿ�ҵ�SO2��ζ�������ڿ������ױ�������������ˮ���ɵ�NaHSO3��Һ�����ԡ�ij�о�С�������ͼ��ʾװ��(ʵ��ǰ�ѳ���װ���ڵĿ���)��ȡNa2S2O5��
(1)װ�����е�Ũ����____(�ܻ���)��ϡ���������ԭ����______________________________________________��
(2)װ��������Na2S2O5����������Ҫ����������ľ������ɲ�ȡ�IJ�����__________��
(3)��Ƽ���Na2S2O5�����ڿ����б��ʵ�ʵ�鷽����_______________��
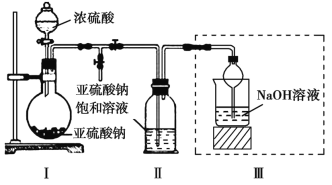
��.Na2S2O3��Һ�������ڲⶨ��Һ��ClO2�ĺ�����ʵ�鷽�����¡�
����1��ȷ��ȡClO2��Һ10.00 mL��ϡ����100 mL��
����2����ȡV1 mLϡ�ͺ��ClO2��Һ����ƿ����������Һ��pH��2������������KI������ҡ�����ڰ�������30���ӡ�(��֪��ClO2+I+H+![]() I2+Cl+H2O δ��ƽ)
I2+Cl+H2O δ��ƽ)
����3���Ե�����Һ��ָʾ������c mol��L1 Na2S2O3��Һ�ζ����յ�������Na2S2O3��ҺV2 mL��(��֪��I2+2S2O32��=2I+ S4O62��)
(1)ȷ��ȡ10.00 mL ClO2��Һ�IJ���������____________��
(2)ȷ���ζ��յ������Ϊ________________________________��
(3)����������������ԭClO2��Һ�����ʵ���Ũ��Ϊ____mol��L1(�ú���ĸ�Ĵ���ʽ��ʾ)��
(4)���в����ᵼ�²ⶨ���ƫ�ߵ���_______________ (����ĸ)��
A��δ�ñ�Ũ�ȵ�Na2S2O3��Һ��ϴ�ζ���
B���ζ�ǰ��ƿ��������ˮ
C���ζ�ǰ�ζ��ܼ��첿�����������ζ���������ʧ
D������ʱ���ζ�ǰ�������ζ�����
���𰸡����ܶ�������������ˮ����ȡ����Na2S2O5�������Թ��У�������ˮ�ܽ⣬�μ����������ᣬ���ٵ����Ȼ�����Һ���а�ɫ�������ɣ���˵��Na2S2O5�����ѱ�����ʽ�ζ��ܵμ����һ��Na2S2O3��Һʱ����Һ�պ�����ɫ��Ϊ��ɫ���ұ���30 s����![]() AC
AC
��������
��.ϡ����ܴ���Ũ���ᣬ���������Ʒ�Ӧ���������������壬��Ϊ��������������ˮ��װ�â�������Na2S2O5���壬ͨ�����˲����õ��þ��壻Na2S2O5�����ڿ������ױ�����ΪNa2SO4�������ᡢ�Ȼ�����Һ������Ʒ���Ƿ�����������ɡ���. ClO2��Һ����ǿ�����ԣ���ȡClO2��Һ����ʽ�ζ��ܣ��ζ��յ�ʱNa2S2O3��Һ����ȫ����ԭ���Ե�����Һ��ָʾ�������ݵ����غ㣬�������Ⱥ͵ⵥ�ʵķ�Ӧ�������¹�ϵ��2ClO2��5I2����Ϸ�Ӧ2Na2S2O3+I2=Na2S4O6+2NaI���õ���ϵʽ��2ClO2��5I2��10Na2S2O3���ɹ�ϵʽ��⼴�ɡ�
��.��1��ϡ����ܴ���Ũ���ᣬ������������Ӧ���������������壬��Ϊ��������������ˮ���ʲ�����ϡ���ᡣ��2��װ�â�������Na2S2O5���壬ͨ�����˲����õ��þ��塣��3��Na2S2O5�����ڿ������ױ�����ΪNa2SO4�������ᡢ�Ȼ�����Һ������Ʒ���Ƿ�����������ɣ�����鷽��Ϊ��ȡ����Na2S2O5�������Թ��У�������ˮ�ܽ⣬�μ��������ᣬ���ٵ����Ȼ�����Һ���а�ɫ�������ɣ���˵��Na2S2O5�����ѱ��ʡ�
��.��1��ClO2��Һ����ǿ�����ԣ�����ȷ��ȡ10.00mLClO2��Һ�IJ�����������ʽ�ζ��ܡ���2���ζ��յ�ʱNa2S2O3��Һ����ȫ����ԭ���Ե�����Һ��ָʾ������Һ��ɫ��ȥ���ζ��յ������Ϊ�μ����һ��Na2S2O3��Һʱ����Һ�պ�����ɫ��Ϊ��ɫ���ұ���30s���䡣��3�����ݵ����غ㣬�������Ⱥ͵ⵥ�ʵķ�Ӧ�������¹�ϵ��2ClO2��5I2����Ϸ�Ӧ2Na2S2O3+I2=
Na2S4O6+2NaI���õ���ϵʽ��2ClO2��5I2��10Na2S2O3��n��S2O32-��=cV2��10-3mol������V1mLClO2����Һ�к��е�ClO2�����ʵ���Ϊ2cV2��10-4mol����10mL��ԭ��Һ����ClO2�����ʵ���Ϊ![]() ��10-2mol������ԭClO2��Һ�����ʵ���Ũ��Ϊ
��10-2mol������ԭClO2��Һ�����ʵ���Ũ��Ϊ![]() ��10-2mol��0.01L=
��10-2mol��0.01L=![]() mol/L����4��A��δ�ñ�Ũ�ȵ�Na2S2O3��Һ��ϴ�ζ��ܣ��ᵼ�±�ҺŨ�ȱ�С����ȥ��Һ�����ƫ�����Բⶨ���ƫ�ߣ�B���ζ�ǰ��ƿ������ˮ����ʵ����Ӱ�죻C���ζ�ǰ�ζ��ܼ��첿�������ݣ��ζ���������ʧ���ᵼ�±�Һ�����ƫ�����Բⶨ���ƫ�ߣ�D���ζ�Ӧ�����Ի������Ի����н��У�����Һ�ʼ��ԣ��ᵼ����ȥ�ı�Һ�����ƫС�����Բⶨ���ƫ�ͣ�E���۲����ʱ���ζ�ǰ���ӣ��ζ����ӣ�������Һ�����������ƫС�����Բⶨ���ƫ�ͣ��ʴ�Ϊ��AC��
mol/L����4��A��δ�ñ�Ũ�ȵ�Na2S2O3��Һ��ϴ�ζ��ܣ��ᵼ�±�ҺŨ�ȱ�С����ȥ��Һ�����ƫ�����Բⶨ���ƫ�ߣ�B���ζ�ǰ��ƿ������ˮ����ʵ����Ӱ�죻C���ζ�ǰ�ζ��ܼ��첿�������ݣ��ζ���������ʧ���ᵼ�±�Һ�����ƫ�����Բⶨ���ƫ�ߣ�D���ζ�Ӧ�����Ի������Ի����н��У�����Һ�ʼ��ԣ��ᵼ����ȥ�ı�Һ�����ƫС�����Բⶨ���ƫ�ͣ�E���۲����ʱ���ζ�ǰ���ӣ��ζ����ӣ�������Һ�����������ƫС�����Բⶨ���ƫ�ͣ��ʴ�Ϊ��AC��

 ��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�
��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�����Ŀ��ijѧ����0.1000 mol/L NaOH��Һ�ζ�δ֪Ũ�ȵ�������Һ�������ɷֽ�Ϊ���¼�����
A.������ˮϴ�ɾ��ζ��ܣ�B. �ô��ⶨ����Һ��ϴ��ʽ�ζ��ܣ�C.����ʽ�ζ���ȡϡ����20.00 mL��ע����ƿ�У������̪��D.��ȡ��ƿ�����ظ�����һ�Σ�E.���ζ����Ƿ�©ˮ��F.ȡ�¼�ʽ�ζ����ñ���NaOH��Һ��ϴ����Һע���ʽ�ζ��ܡ�0���̶�����2��3 cm�����ٰѼ�ʽ�ζ��̶ܹ��ã�����Һ������0���̶Ȼ�0���̶����£�
G.����ƿ���ڵζ������棬ƿ�µ�һ�Ű�ֽ���ߵα�ҡ����ƿֱ���ζ��յ㣬���µζ���Һ�����ڿ̶ȡ����������գ�
��1����ȷ������˳����(�������ĸ��д)_____________��
��2������F��Ӧ��ѡ��ͼ��________�ζ���(����)��
��3���ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�________��
A. �ζ�����Һ��ı仯 B. ��ƿ����Һ��ɫ�ı仯
�ζ��յ���Һ��ɫ�ı仯��____________________________��
��4���ζ���������ʾ��
�ζ����� | ������Һ���/mL | ����Һ�����/mL | |
�ζ�ǰ�̶� | �ζ���̶� | ||
1 | 20 | 1.02 | 21.03 |
2 | 20 | 2.00 | 25.00 |
3 | 20 | 0.60 | 20.60 |
�ζ������ϴ���ǵ�________��ʵ�飬����������Ŀ���ԭ����________��
A. ��ʽ�ζ�����װҺǰδ�ñ�NaOH��Һ��ϴ2��3��
B. �ζ���ʼǰ��ʽ�ζ��ܼ��첿�������ݣ��ڵζ��յ����ʱδ��������
C. �ζ���ʼǰ��ʽ�ζ��ܼ��첿��û�����ݣ��ڵζ��յ����ʱ���ּ��첿��������
D. �ﵽ�ζ��յ�ʱ��������Һ��Һ����͵����
E. �ζ������У���ƿҡ����̫���ң�������ЩҺ�ηɽ�����
��5���������Ũ��Ϊ________mol/L��
��6�����ȷ��ȡ20.00 mL 0.1000 mol/L NaOH��Һ����ƿ�У������ָ̪ʾ����Ȼ����δ֪Ũ�ȵ�����(װ����ʽ�ζ�����)�ζ����Ƿ�Ҳ�ɲⶨ����������ʵ���Ũ�ȣ�________(��ǡ���)��