��Ŀ����
��10�֣�
�������зḻ�ĵ⡣Ϊ�˴Ӻ�������ȡ�⣬ij�о���ѧϰС����Ʋ�����������ʵ�飺
����д���пհף�
��1����������պ���ʱ������Ҫ���Ǽ��⣬����Ҫ�õ���������
��������������ѡ��������������ñ����ĸ��д�ڿհ״���
A���ձ� B������ C�������� D�������� E���ƾ��� F��������
��2������۵�ʵ����������� �������Ŀ���ǴӺ��ⱽ��Һ�з������ͻ��ձ����ò����ʵ����������� ��
��3��������У�ijѧ��ѡ���ñ�����ȡ���������
��
��4�������һ�ּ�����ȡ����ˮ��Һ���Ƿ��е��ʵ�ļ���
��
��1��B��D��E
��2�����ˡ�����
��3������ˮ�����ܣ����ڱ��е��ܽ�����Ա���ˮ�д�
��4����С�Թ�ȡ������Һ��������²㣨ˮ�㣩��Һ�����뼸�ε�����Һ�������ɫ֤�������ⵥ��
����:

 ��ĩ1�����ʽ���������ϵ�д�
��ĩ1�����ʽ���������ϵ�д� ������������Ԫ��֮һ������ֲ���纣���������к��зḻ���Ե�������ʽ���ڵĵ�Ԫ�أ������к��зḻ�ĵ⣮Ϊ�˴Ӻ�������ȡ�⣬ij�о���ѧϰС����Ʋ�����������ʵ�飺
������������Ԫ��֮һ������ֲ���纣���������к��зḻ���Ե�������ʽ���ڵĵ�Ԫ�أ������к��зḻ�ĵ⣮Ϊ�˴Ӻ�������ȡ�⣬ij�о���ѧϰС����Ʋ�����������ʵ�飺
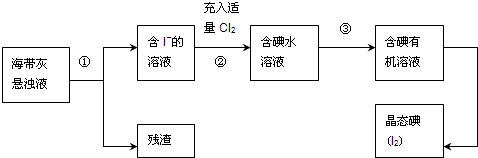
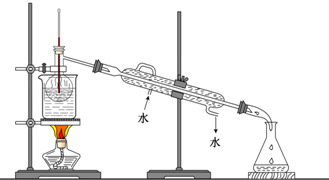

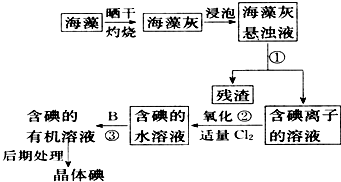 ����ֲ���纣���������к��зḻ�ĵ�Ԫ�أ���Ԫ�������ӵ���ʽ���ڣ�ʵ���дӺ�����ȡ���������ͼ��ʾ��
����ֲ���纣���������к��зḻ�ĵ�Ԫ�أ���Ԫ�������ӵ���ʽ���ڣ�ʵ���дӺ�����ȡ���������ͼ��ʾ��