��Ŀ����
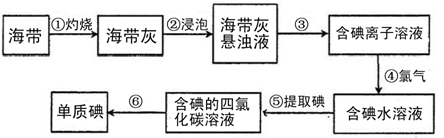
�����к��зḻ�ĵ⣬Ϊ�˴Ӻ�������ȡ�⣬ij�о���ѧϰС����Ʋ�����������ʵ�飺
��1������������õ�1mol/L��ϡ����100mL������98%���ܶ�Ϊ1.84g/cm3��Ũ���������ƣ���Ӧ����Ͳ��ȡŨ����������
��2������۵�ʵ�����������
��3���������ѡ���ñ������þƾ�����ȡ���������
��4���������ϣ���С��ͬѧ֪�������еĵ�Ԫ����Ҫ���л��⻯�����ʽ���ڣ�����С��ʵ��ʱ�Ƶú���������Ϊag����ȡ��Ĺ����е���ʧ��m%�����õ����ʵ������Ϊbg����С��ͬѧ��õĺ����е�ĺ���Ϊ���ú�a��b��m��ʽ�ӱ�ʾ��
%��
%
%��
%��

��1������������õ�1mol/L��ϡ����100mL������98%���ܶ�Ϊ1.84g/cm3��Ũ���������ƣ���Ӧ����Ͳ��ȡŨ����������
5.4mL
5.4mL
����2������۵�ʵ�����������
����
����
�������Ŀ���ǴӺ��ⱽ��Һ�з�������ʵ�ͻ��ձ����ò����ʵ���������������
����
����3���������ѡ���ñ������þƾ�����ȡ���������
�ƾ�����ˮ����������ˮ
�ƾ�����ˮ����������ˮ
����4���������ϣ���С��ͬѧ֪�������еĵ�Ԫ����Ҫ���л��⻯�����ʽ���ڣ�����С��ʵ��ʱ�Ƶú���������Ϊag����ȡ��Ĺ����е���ʧ��m%�����õ����ʵ������Ϊbg����С��ͬѧ��õĺ����е�ĺ���Ϊ���ú�a��b��m��ʽ�ӱ�ʾ��
| b |
| a(1-m) |
| 100b |
| a(1-m%) |
| b |
| a(1-m) |
| 100b |
| a(1-m%) |

��������1��������Һϡ��ǰ�����ʵ����ʵ��������������Ũ������Һ�����������Ũ��������ѡȡ��Ͳ��ע����ѡ��Ͳ���ݻ�Ӧ�Դ��ڻ������ȡ��Һ�������
��2����������Һ���ù��ˣ����뻥�ܵ�����Һ��������
��3��������ȡʱ��ȡ����ѡ��Ӧ����������������������ȡ���е��ܽ�ȱ���ԭ�ܼ���Ҫ����ȡ����ԭ�ܼ������ܣ���ȡ�������ʲ���Ӧ��
��4��������ܵĵⵥ�ʵ������������е������������
��2����������Һ���ù��ˣ����뻥�ܵ�����Һ��������
��3��������ȡʱ��ȡ����ѡ��Ӧ����������������������ȡ���е��ܽ�ȱ���ԭ�ܼ���Ҫ����ȡ����ԭ�ܼ������ܣ���ȡ�������ʲ���Ӧ��
��4��������ܵĵⵥ�ʵ������������е������������
����⣺��1��Ũ��������ʵ���Ũ��Ϊ
=
mol/L=18.4mol/L������ҪŨ����������Һ�����ΪV��0.2mol/L��0.5L=18.4mol/L��V��V=0.0054L=5.4mL��
�ʴ�Ϊ��5.4mL��
��2��������Ƿ�������Һ�壬��ʵ�����Ϊ���ˣ������Ŀ���ǴӺ��ⱽ��Һ�з�������ʵ�ͻ��ձ��������û��ܵ�����Һ��ķе㲻ͬ�����룬��ʵ�����Ϊ����
�ʴ�Ϊ�����ˣ�����
��3���ƾ���ˮ���ܶ���������ˮ���ʴ�Ϊ���ƾ�����ˮ����������ˮ��
��4���ܵĵⵥ�ʵ�����Ϊ
�����е����������Ϊ
%���ʴ�Ϊ��
%��
%��
| 103��w |
| M |
| 103��1.84��98% |
| 98 |
�ʴ�Ϊ��5.4mL��
��2��������Ƿ�������Һ�壬��ʵ�����Ϊ���ˣ������Ŀ���ǴӺ��ⱽ��Һ�з�������ʵ�ͻ��ձ��������û��ܵ�����Һ��ķе㲻ͬ�����룬��ʵ�����Ϊ����
�ʴ�Ϊ�����ˣ�����
��3���ƾ���ˮ���ܶ���������ˮ���ʴ�Ϊ���ƾ�����ˮ����������ˮ��
��4���ܵĵⵥ�ʵ�����Ϊ
| b |
| (1-m%) |
| 100b |
| a(1-m%) |
| b |
| a(1-m) |
| 100b |
| a(1-m%) |
���������⿼����һ�����ʵ���Ũ����Һ�����ƣ������е����ȡ���Ѷ����У�����ѧ�����ʵ�������������ע��������ȡ��Һ�������ȷѡȡ��Ͳ��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ