��Ŀ����
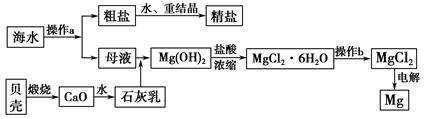
��±�Ǻ�ˮ�����ᾧ�����ʳ�κ��ĸҺ����±�к��н϶��NaCl��MgCl2��KBr��MgSO4�����ʡ�������������Ŀǰ�Ӻ�ˮ����ȡ��ij��÷�����������ͼ��ʾ��
����˵����ȷ���� (����)��

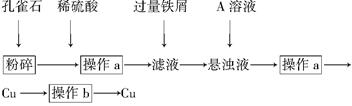
����˵����ȷ���� (����)��
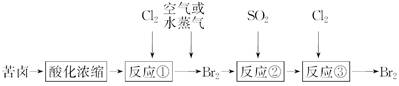
| A����Ӧ�٢۵ķ���ʽ��ͬ����Ŀ��Ҳ��ȫ��ͬ |
| B����ò��÷�Һ�ķ����ӷ�Ӧ�ۺ����Һ�з���������� |
| C��ͼʾ�Ĺ���������һ��û���κ�ȱ�ݵij��켼�� |
| D��������������Ӧ���ܴӿ�±��ұ��������þ |
D
�ٷ�Ӧ����ʽ��2KBr��Cl2=2KCl��Br2���۷�Ӧ����ʽ��2HBr��Cl2=2HCl��Br2����Ӧ��Ļ��Һͨ����Һ����ʵ��Br2����ȡ�ģ����Բ�������

��ϰ��ϵ�д�
�����Ŀ