��Ŀ����
A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��ʾ��

��������²�ͬ����ش�
��1����A��B��C����ɫ��Ӧ���ʻ�ɫ��ˮ��Һ��Ϊ���ԡ�
��A�������еĻ�ѧ����_____________��
�ڽ�4.48 L����״���£�Xͨ��100mL 3 mol��L A��ˮ��Һ����Һ������Ũ���ɴ�С��˳�� Ϊ_______________________________________��
Ϊ_______________________________________��
����Ȼ���д���B��C��H2O��һ�������ᾧ���ɵĹ��塣ȡһ�����ù�������ˮ���100mL��Һ����������н��������ӵ�Ũ��Ϊ0.5 mol��L����ȡ��ͬ�����Ĺ�����������أ�ʣ����������Ϊ__________��
��2����AΪ��̬�ǽ������ʣ�A��Xͬ���ڣ����³�ѹ��CΪ��ɫ���壬B�����и�ԭ��������Ϊ8e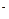 �ṹ��
�ṹ��
�������й�B���ʵ�������ȷ����
a��B�ķ���ʽΪAX b��BΪ���ۻ�����
c��B���ӳ������� d��B�����ȶ��������X����κ����ʷ�����ѧ��Ӧ
��C��ˮ���ҷ�Ӧ���������ֳ����ᣬ��Ӧ�Ļ�ѧ����ʽΪ ��

��������²�ͬ����ش�
��1����A��B��C����ɫ��Ӧ���ʻ�ɫ��ˮ��Һ��Ϊ���ԡ�
��A�������еĻ�ѧ����_____________��
�ڽ�4.48 L����״���£�Xͨ��100mL 3 mol��L A��ˮ��Һ����Һ������Ũ���ɴ�С��˳��
 Ϊ_______________________________________��
Ϊ_______________________________________������Ȼ���д���B��C��H2O��һ�������ᾧ���ɵĹ��塣ȡһ�����ù�������ˮ���100mL��Һ����������н��������ӵ�Ũ��Ϊ0.5 mol��L����ȡ��ͬ�����Ĺ�����������أ�ʣ����������Ϊ__________��
��2����AΪ��̬�ǽ������ʣ�A��Xͬ���ڣ����³�ѹ��CΪ��ɫ���壬B�����и�ԭ��������Ϊ8e
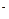 �ṹ��
�ṹ���������й�B���ʵ�������ȷ����
a��B�ķ���ʽΪAX b��BΪ���ۻ�����
c��B���ӳ������� d��B�����ȶ��������X����κ����ʷ�����ѧ��Ӧ
��C��ˮ���ҷ�Ӧ���������ֳ����ᣬ��Ӧ�Ļ�ѧ����ʽΪ ��
��1�������Ӽ� ���ۼ� �� c(Na+)>c(HCO
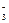 )>c(CO
)>c(CO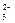 ) >c(OH
) >c(OH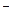 ) >c( H+ )����2.65 g
) >c( H+ )����2.65 g��2���� bc ��PCl5+4H2O=H3PO4+5HC1
��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ

 ���������
���������
 AΪ��ɫ�������ʣ�������Ϊ1.92g���������B�ڳ����·�Ӧ����������ȫ������ɫ����C�ռ�������Ȼ����ˮ�У���ͨ���״����VmL������
AΪ��ɫ�������ʣ�������Ϊ1.92g���������B�ڳ����·�Ӧ����������ȫ������ɫ����C�ռ�������Ȼ����ˮ�У���ͨ���״����VmL������ ǡ��ʹC������ȫת��ΪB����V=________��
ǡ��ʹC������ȫת��ΪB����V=________��