��Ŀ����
����Ŀ����0.1 mol��þ�������������100 mL 2mol/LH2SO4��Һ�У�Ȼ���ٵμ�1 mol/L NaOH��Һ����ش�

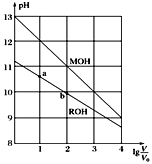
�����ڵμ�NaOH��Һ�Ĺ����У���������m�����NaOH��Һ�����V�仯����ͼ��ʾ����V1��160mLʱ���������ĩ��n(Mg)=______________mol��V2��______________mL��
�����ڵμ�NaOH��Һ�����У���ʹMg2����Al3���պó�����ȫ�������NaOH��Һ�����V(NaOH)_________mL��
�����������Ϊ0.1 mol������Mg�۵����ʵ�������Ϊa����100 mL 2 mol/L�������ܽ�˻������ټ���450 mL 1mol/L��NaOH��Һ�����ó�������Al(OH)3�������������a��ȡֵ��Χ�ǣ�______________��
���𰸡�0.06 mol 440 mL 400 mL ![]() ��a��1
��a��1
��������
����ͼ���֪��V1 mL��NaOH��Һ�������H2SO4��Ӧ�ģ�����Mg��Al��Ӧ��ΪH2SO4��
0.2mol��0.16mol��![]() ��0.12mol������n(Mg)��x mol��n(Al)��y mol�����з����飺
��0.12mol������n(Mg)��x mol��n(Al)��y mol�����з����飺
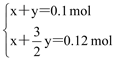 ����ã�x��0.06 mol��y��0.04 mol��
����ã�x��0.06 mol��y��0.04 mol��
������V2 mL NaOH��Һʱ���õ�Mg(OH)2����NaAlO2����ϵʽΪ��
Mg2����2OH����Al3����4OH������V2��160 mL��![]() ��440 mL��
��440 mL��
�Ƶ�Mg2����Al3���պó�����ȫʱ����Һ��ֻ��Na2SO4����ϵʽΪ2NaOH��H2SO4����V(NaOH)��![]() ��400 mL��
��400 mL��
�ǵ����ó�������Al(OH)3ʱ��NaOH��Һ��������Ӧ�����ɵ�����Ϊ��Na2SO4��NaAlO2��
�����NaԪ���غ㣺n(NaOH)��0.45 L��1mol/L��0.1 L��2 mol/L��2��0.1��(1��a)mol��
���a��![]() ���������������a��ȡֵ��Χ��
���������������a��ȡֵ��Χ��![]() ��a��1��
��a��1��
�𰸣���0.06 mol��440 mL�� ��400 mL�� ��![]() ��a��1��
��a��1��

����Ŀ��������![]() ������
������![]() ���
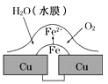
���![]() ��ˮ��Һ�����һ��ʱ����ټ���
��ˮ��Һ�����һ��ʱ����ټ���![]() ����ʹ��Һ�ָ������ǰ��״̬�����������һ����
����ʹ��Һ�ָ������ǰ��״̬�����������һ����
|
|
|
| |
A |
|
|
|
|
B |
|
|
|
|
C |
|
|
|
|
D |
|
|
|
|
A. A B. B C. C D. D