��Ŀ����
ij�о���ѧϰС������ữ����������Һ����I�����ڵĺ����Բ�������Ȥ��ͬѧ�Ǹ��ݱ�������˼��������·�����Ʋ�������ʵ��̽����
[�������]
����1�����ɵ�AgI������ˮ�����ܱ�HNO3������
����2��HNO3�������ԣ��ܽ�I��������I2��
[���ʵ�鷽������֤����]
��1����ͬѧ��KI��Һ�еμ����ữ��AgNO3��Һ�����л�ɫ�������ɡ���֤�˼���1��������д���йػ�ѧ����ʽ ��
��2����ͬѧ���ʵ����֤2�����������±������ݡ�
| ʵ�鲽�裨��Ҫ��д����������̣� | Ԥ������ͽ��� |
| | ����Һ����������2������ ����Һ������������2�������� |
| ���� |
[���������]
��3����ͬѧ��֤�˼���1����������ͬѧ��֤�˼���2���������ͬѧ�ɴ˵ó��Ľ��ۣ������ữ��AgNO3����ʹI��������Ҳ������I������ͬ���ͬѧ�Ľ����𣬲��������ɣ� ��
[˼���뽻��]
��3���о���ѧϰС��ͬѧ�����о�Fe3+�ܷ�����I�������⣬���������ַ���������ʵ����֤��
����һ��
��������
������֤��������д����Ҫ���ӷ���ʽ ��
(12�֣�ÿ��2��)
��1��KI+AgNO3=AgI��+KNO3
��2��ȡ��ͬѧ���õ������ữ��AgNO3�μӵ�KI��������Һ��
��3����ͬ�⣬�����ữ��AgNO3���������ᣬ����֤�������Ƿ�������I-
��4������һ����FeCl3��Һ�еμӼ���KI��������Һ������Һ�Ƿ����
����������KI��KSCN��Һ�еμӼ���FeCl3��Һ������Һ�Ƿ��ȱ�����ɫ2Fe3++2I-=I2+2Fe2+
���������������2�����������ǿ�����Ժ͵�ʹ���۱���ɫ�����ԣ���4���������֤ʵ�鷽��ʱҪע�����һ�����������ǵ�����Ϊ��֤�������Ƿ���I2���Ƿ���Fe3+��
���㣺����ʵ�鷽������ƺ����ۣ����鿼�����ʵ��ͽ���ʵ�����۵�������

 ��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�ijNa2CO3��Ʒ�л���һ������Na2SO4 (��������ᾧˮ����ij��ѧ��ȤС��������ַ����ⶨ����Ʒ��Na2CO3�������������Իش��������⡣
����һ���������з������Na2CO3���������Ĝy��
(1)�����ۺܵ͢����Ʒֱ�Ϊ_______��
(2)���������١����У�ʹ�õ�����������______(��������)��
(3)�жϲ����ڷ���ɵķ�����______
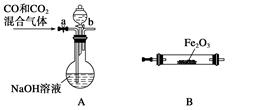
��������������ͼʵ��װ�ã��г�������ʡ�ԣ�.ѡ�������Լ�: a.Ũ����b.����NaHCO3��ҺC.6mol/L����D.2mol/L����, e.��ʯ��f. ��ˮCaCl2,�y����Ʒ��Na2CO3,������������
(4)��д���пո�
| ���� | �Լ� | ������Լ���Ŀ�� |
| A | | �������ʱϴȥCO2 |
| B | | ʹ��Ʒ��ַ�Ӧ�ų����� |
| C | a | |
| D | e | �������CO2 |
| E | e | |
��ˮ��Դ�����ð�����ˮ�ĵ��������·����У����ܵ�����ˮ����
| A������ | B�����˷� | C���������� | D�����ӽ����� |
 3Na2S2O3+CO2��ij�о�С����ʵ����ģ��ù�ҵԭ���Ʊ�Na2S2O3������ʵ��װ������:
3Na2S2O3+CO2��ij�о�С����ʵ����ģ��ù�ҵԭ���Ʊ�Na2S2O3������ʵ��װ������:




 Mg��OH��2
Mg��OH��2 MgCl2��Һ��MgCl2��6H2O��MgCl2
MgCl2��Һ��MgCl2��6H2O��MgCl2 Mg
Mg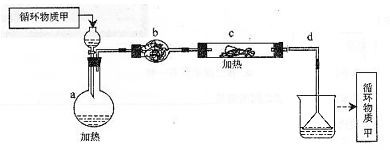
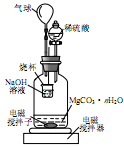
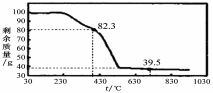

 �����£�
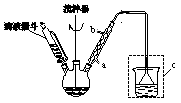
�����£�
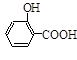 ��ˮ���ᣩ��SOCl2��
��ˮ���ᣩ��SOCl2��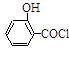 ��ˮ�����ȣ���HCl����SO2��
��ˮ�����ȣ���HCl����SO2��  ]���¶ȿ�����100�����ң����Ͻ��衣
]���¶ȿ�����100�����ң����Ͻ��衣