��Ŀ����
����Ŀ�������仯������������������������Ҫ����;����֪: Sn���۵�Ϊ231����Sn2+��ˮ�⡢�ױ�������SnCl4����ˮ�⡢�۵�Ϊ-33�桢�е�Ϊ114�档��ش���������:
��1��Ԫ������ͬ����̼����������3,����ԭ������Ϊ_________��
��2�����������������������۴��Ȳⶨ(��֪�������ԭ������Ϊ119): ��ȡ1.19g��������ϡ������(���ʲ����뷴Ӧ),ʹSn ��ȫת��ΪSn2+���ڼ��������Fe2(SO4)3������0.1000mol/LK2Cr2O7��Һ�ζ�(������Cr��+3��),����20.00mL��������м���Fe2(SO4)3��������________����������Ʒ��������������Ϊ_________�����ڵζ��յ�ʱ���Ӷ�������ᵼ�²�õ���Ʒ��������������________(����ƫ����ƫ����������Ӱ����)��
��2�����ڶ�����ҵ����������(SnSO4)���Ʊ�·������:

�ٲ���I����Sn�۵����ã�____________��������ҺpH��
�ڲ���II�õ��IJ����������ձ���____________��
�۲���III����SnO�����ӷ���ʽ: ____________��
�ܲ���IV�м���SnO�Ƿ�ϴ�Ӹɾ��IJ�����____________��֤����ϴ����
�ݲ���V��������Ϊ____________ ��____________���ˡ�ϴ�ӡ����¸��
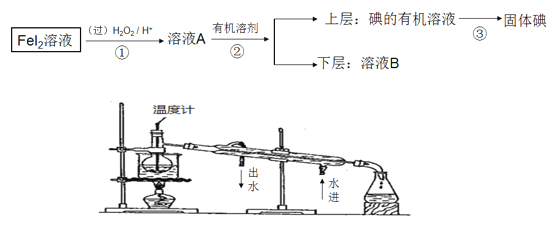
(4)ʵ����������ͼװ���Ʊ�����SnCl4(�г�װ����),��װ�ô�������ȱ��,�Ľ�������____________��

���𰸡� 50 ��Sn2+ȫ������ΪSn4+ 60% ƫ�� ��ֹSn2+������ΪSn4+�� ©��(����ͨ©��) ������ Sn2++2HCO3-= SnO��+2CO2��+ H2O ȡ���һ����Һ(ˮ��Һ) �������Թ��У����εμ��������ᡢ������������Һ���۲쵽�ް�ɫ���� ����(����) Ũ������ȴ�ᾧ ��A��Bװ�ü���������ʢ�б���ʳ��ˮ��Ũ�����ϴ��ƿ
�������������������1��Ԫ������ͬ����̼����������3���������ڵ������ڵڢ�A����
��2��Fe3+�ܰ�Sn2+����ΪSn4+�����ݹ�ϵʽ����������Ʒ�������������������ڵζ��յ�ʱ���Ӷ���������K2Cr2O7��Һ�����ƫС����3����Sn2+�ױ���������ΪSn4+���ڲ���II�ǹ��ˣ��۲���III Sn2+��HCO3-����˫ˮ�ⷴӦ���ܲ���IV�м���SnO�Ƿ�ϴ�Ӹɾ��IJ����Ǽ���ϴ��Һ���Ƿ���������������������Һ����Ũ�������½ᾧ�ɵ����������壻(4)����ͼʾװ����ȡ�����������Ȼ����ˮ���������ɵ�SnCl4������ˮ�ⷴӦ��
��������1��Ԫ������ͬ����̼����������3���������ڵ������ڵڢ�A�壬��������ԭ��������6+8+18+18=50��
��2��Fe3+�ܰ�Sn2+����ΪSn4+��������������Sn2+ȫ������ΪSn4+�����ݹ�ϵʽ3Sn ~~3 Sn2+~~3 Fe2(SO4)3~~ K2Cr2O7��n(K2Cr2O7)= 0.02L��0.1mol/L=0.002mol��n(Sn)= 0.002mol��3=0.006mol��������Ʒ��������������Ϊ![]()
���ڵζ��յ�ʱ���Ӷ���������K2Cr2O7��Һ�����ƫС��������������ƫ������3����Sn2+�ױ���������ΪSn4+���������ۿ��Է�ֹSn2+������ΪSn4+���ڲ���II�ǹ�����ʹ���������ձ���©�������������۲���III Sn2+��HCO3-����˫ˮ�ⷴӦ����Ӧ�����ӷ���ʽ��Sn2++2HCO3-=SnO��+2CO2��+H2O���ܲ���IV�м���SnO�Ƿ�ϴ�Ӹɾ��IJ����Ǽ���ϴ��Һ���Ƿ���������������������ȡ���һ����Һ(ˮ��Һ)�������Թ��У����εμ��������ᡢ������������Һ���۲쵽�ް�ɫ��������ϴ�Ӹɾ�������������Һ����Ũ�������½ᾧ�ɵ����������壬�������������IJ�������Ϊ����Ũ�������½ᾧ�����ˡ�ϴ�ӡ����¸�����(4)����ͼʾװ����ȡ�����������Ȼ����ˮ���������ɵ�SnCl4������ˮ�ⷴӦ���Ľ���������A��Bװ�ü���������ʢ�б���ʳ��ˮ��Ũ�����ϴ��ƿ��ȥ�Ȼ����ˮ������

 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�