��Ŀ����
Ԫ�����ڱ���Ԫ����������ѧϰ���о�������ʵ�����к���Ҫ�����ã��±��г��ˢ١������Ԫ�������ڱ��е�λ�ã���ش�
��1�������Ԫ���У���Ԫ�ط��ţ����� �����л�ѧ��������õ��� ���ݺ͢��γɸۻ�����ĵ���ʽ
��2������ЩԪ�ص�����������Ӧ��ˮ�����У�������ǿ���ǣ��ѧʽ�� ������ǿ���� �����Ե��� ��д������֮�����Ӧ�Ļ�ѧ����ʽ ��
��3���١��ڡ�������Ԫ�ذ�ԭ�Ӱ뾶�ɴ�С��˳������Ϊ ����Ԫ�ط��ţ���
��4���õ���ʽ��ʾ��Ԫ�����Ԫ���γɻ�����Ĺ��� ��
��5���ڢٺ͢��л�ѧ��������õ���
��6���ڢߺ͢��л�ѧ��������õ��� ���û�ѧʵ��֤���ķ��������ӷ���ʽ������ ���ӷ���ʽ ��
| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� |
��2������ЩԪ�ص�����������Ӧ��ˮ�����У�������ǿ���ǣ��ѧʽ��
��3���١��ڡ�������Ԫ�ذ�ԭ�Ӱ뾶�ɴ�С��˳������Ϊ
��4���õ���ʽ��ʾ��Ԫ�����Ԫ���γɻ�����Ĺ���
��5���ڢٺ͢��л�ѧ��������õ���
��6���ڢߺ͢��л�ѧ��������õ���
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ã���֪��ΪNa����Ϊk����ΪMg����ΪAl����ΪC����ΪO����ΪCl����ΪBr����ΪAr��
��1��ϡ�����廯ѧ��������ã��ݺ͢��γɸۻ�����ΪCO2��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ�
��2��������Խǿ������������Ӧˮ����ļ���Խǿ���ǽ�����Խǿ����ۺ����������Խǿ�������������������������
��3��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����
��4����Ԫ�����Ԫ���γɻ�����ΪMgBr2����þ�����������ӹ��ɣ���Mgԭ�ӡ���ԭ�ӵ���ʽ��ʾ���γɣ�
��5��ͬ�������϶��½�������������Խǿ������Խ���ã�
��6��ͬ�������϶��·ǽ����Լ������ǽ�����Խǿ����ѧ����Խ���ã��������õ���֮�����û�������֤��
��1��ϡ�����廯ѧ��������ã��ݺ͢��γɸۻ�����ΪCO2��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ�
��2��������Խǿ������������Ӧˮ����ļ���Խǿ���ǽ�����Խǿ����ۺ����������Խǿ�������������������������
��3��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����
��4����Ԫ�����Ԫ���γɻ�����ΪMgBr2����þ�����������ӹ��ɣ���Mgԭ�ӡ���ԭ�ӵ���ʽ��ʾ���γɣ�
��5��ͬ�������϶��½�������������Խǿ������Խ���ã�
��6��ͬ�������϶��·ǽ����Լ������ǽ�����Խǿ����ѧ����Խ���ã��������õ���֮�����û�������֤��
���
�⣺��Ԫ�������ڱ���λ�ã���֪��ΪNa����Ϊk����ΪMg����ΪAl����ΪC����ΪO����ΪCl����ΪBr����ΪAr��
��1������Ԫ���У���ΪCԪ�أ�ϡ������Ar��ѧ��������ã��ݺ͢��γɸۻ�����ΪCO2��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ�����ʽΪ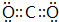 ���ʴ�Ϊ��C��Ar��
���ʴ�Ϊ��C��Ar��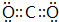 ��
��
��2������Ԫ���У�K�Ľ�������ǿ����KOH�ļ�����ǿ������Ԫ�غ���Cl�ǽ�������ǿ������ۺ�����HClO4������Խǿ��Al��OH��3�����������������֮�䷢���ķ�Ӧ����ʽΪ��KOH+HClO4=KClO4+H2O��KOH+Al��OH��3=KAlO2+2H2O��3HClO4+Al��OH��3=Al��ClO4��3+3H2O��
�ʴ�Ϊ��KOH��HClO4��Al��OH��3��KOH+HClO4=KClO4+H2O��KOH+Al��OH��3=KAlO2+2H2O��3HClO4+Al��OH��3=Al��ClO4��3+3H2O��
��3��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����ԭ�Ӱ뾶��K��Na��Mg���ʴ�Ϊ��K��Na��Mg��
��4����Ԫ�����Ԫ���γɻ�����ΪMgBr2����þ�����������ӹ��ɣ���Mgԭ�ӡ���ԭ�ӵ���ʽ��ʾ���γɹ���Ϊ�� ��
��
�ʴ�Ϊ�� ��
��
��5��ͬ�������϶��½�������������Խǿ������Խ���ã��ʻ�����K��Na���ʴ�Ϊ��K��
��6��ͬ�������϶��·ǽ����Լ������ǽ�����Խǿ����ѧ����Խ���ã��ʻ�����Cl��Br���������õ���֮�����û�������֤��ʵ�鷽��Ϊ��ȡ�����廯����Һ������ˮ����Һ��Ϊ�Ȼ�ɫ����ǽ�����Cl��Br����Ӧ���ӷ���ʽΪ��Cl2+2Br-=2Cl-+Br2��
�ʴ�Ϊ��Cl��ȡ�����廯����Һ������ˮ����Һ��Ϊ�Ȼ�ɫ����ǽ�����Cl��Br��Cl2+2Br-=2Cl-+Br2��
��1������Ԫ���У���ΪCԪ�أ�ϡ������Ar��ѧ��������ã��ݺ͢��γɸۻ�����ΪCO2��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ�����ʽΪ
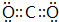 ���ʴ�Ϊ��C��Ar��
���ʴ�Ϊ��C��Ar��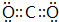 ��
����2������Ԫ���У�K�Ľ�������ǿ����KOH�ļ�����ǿ������Ԫ�غ���Cl�ǽ�������ǿ������ۺ�����HClO4������Խǿ��Al��OH��3�����������������֮�䷢���ķ�Ӧ����ʽΪ��KOH+HClO4=KClO4+H2O��KOH+Al��OH��3=KAlO2+2H2O��3HClO4+Al��OH��3=Al��ClO4��3+3H2O��
�ʴ�Ϊ��KOH��HClO4��Al��OH��3��KOH+HClO4=KClO4+H2O��KOH+Al��OH��3=KAlO2+2H2O��3HClO4+Al��OH��3=Al��ClO4��3+3H2O��
��3��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����ԭ�Ӱ뾶��K��Na��Mg���ʴ�Ϊ��K��Na��Mg��
��4����Ԫ�����Ԫ���γɻ�����ΪMgBr2����þ�����������ӹ��ɣ���Mgԭ�ӡ���ԭ�ӵ���ʽ��ʾ���γɹ���Ϊ��
 ��
���ʴ�Ϊ��
 ��
����5��ͬ�������϶��½�������������Խǿ������Խ���ã��ʻ�����K��Na���ʴ�Ϊ��K��
��6��ͬ�������϶��·ǽ����Լ������ǽ�����Խǿ����ѧ����Խ���ã��ʻ�����Cl��Br���������õ���֮�����û�������֤��ʵ�鷽��Ϊ��ȡ�����廯����Һ������ˮ����Һ��Ϊ�Ȼ�ɫ����ǽ�����Cl��Br����Ӧ���ӷ���ʽΪ��Cl2+2Br-=2Cl-+Br2��
�ʴ�Ϊ��Cl��ȡ�����廯����Һ������ˮ����Һ��Ϊ�Ȼ�ɫ����ǽ�����Cl��Br��Cl2+2Br-=2Cl-+Br2��
���������⿼��Ԫ�����ڱ���Ԫ���������ۺ�Ӧ�ã��Ѷ��еȣ�ע�������õ���ʽ��ʾ���ʻ�ѧ���γɣ����ս����ԡ��ǽ�����ǿ���ȽϷ�����

��ϰ��ϵ�д�
 ��ѧ��ʦ����ϵ�д�
��ѧ��ʦ����ϵ�д�
�����Ŀ
�������Ʒֱ�Ͷ���������ʵ�ˮ��Һ�У������������ɫ�������ǣ�������
| A��NaCl |
| B��K2SO4 |
| C��CuCl2 |
| D��MgCl2 |
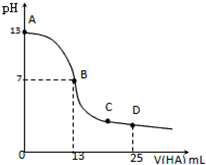 �����£���25mL 0.1mol/L MOH��Һ����μ���0.2mol/L HA��Һ��������ͼ��ʾ������仯���Բ��ƣ�������˵��������ǣ�������
�����£���25mL 0.1mol/L MOH��Һ����μ���0.2mol/L HA��Һ��������ͼ��ʾ������仯���Բ��ƣ�������˵��������ǣ�������| A��MOH�ĵ��뷽��ʽΪMOH=M++OH- |
| B��MOH��HAǡ����ȫ��Ӧʱ����Һ�ʼ��ԣ�������A-+H2O=HA+OH-����ʱ�������Һ����ˮ�������c��H+������0.2mol/L HA��Һ����ˮ�������c��H+�� |
| C�������Һ����B�㣺c��M+��=c��A-����c��H+��=c��OH-������C�㣺c��A-����c��M+����c��H+����c��OH-�� |
| D����Һ��D��ʱ c��A-��+c��HA��=2c��M+��������ʱ���pH=3����c��HA��+c��H+��=0.05+10-11mol/L |
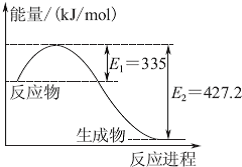 ��1����֪��C��s��+O2��g���TCO2��g����H=-393.5kJ/mol
��1����֪��C��s��+O2��g���TCO2��g����H=-393.5kJ/mol