��Ŀ����
��ʽ̼��ͭ����������ľ�ķ������ȡ���ҵ�Ͽ���ʴ�̷�Һ������һ����Cu2+��H+��SO42-��H2O2����Fe3+���Ʊ��ߴ��ȼ�ʽ̼��ͭ�����Ʊ��������£�
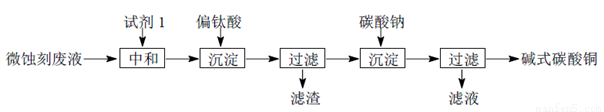
��1���Լ�1���ѡ��________________��
a��Fe2O3 b��CaCO3 C��CuO
��2������Na2CO3����ǰ���ȥFe3+������Fe2(SO4)3����Na2CO3��Ӧ������һ�����壬ͬʱ����NaFe3(OH)6(SO4)2������Ӱ���Ʒ������д���÷�Ӧ�Ļ�ѧ����ʽ______________________��
��3������Na2CO3����ʱ����ӦҺpH�Բ�Ʒ��SO42-�ĺ����Լ���ӦҺ��Cu2+����Ч�ʵ�Ӱ������ͼ��ʾ���ڶ��γ���ʱ��ҺpH��ÿ�����___________________________��
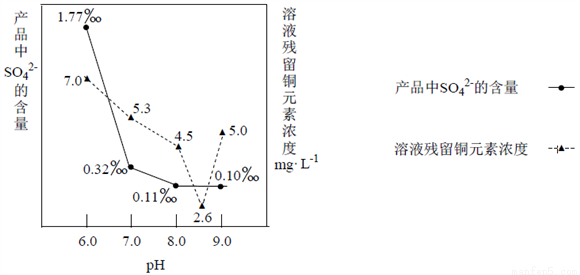
��4�����Ƶõļ�ʽ̼��ͭ����ɿɱ�ʾΪ��xCuCO3��yCu(OH)2��zH2O��Ҫ�ⶨ����ɣ���ͬѧ��Ƶ�ʵ�鷽������Ҫ����������裺�ٳ�ȡ12.0�˵���Ʒ���ڸ��·ֽ⣻�۲������CO2������Ϊ2.2�ˣ��ܲ������ˮ����������Ϊ1.8�ˣ��ݳ�������CuO������Ϊ8.0�ˡ�����ͬѧ��Ϊ��������⣬ʵ��ֻ��ⶨ�ĸ����еIJ������ݼ��ɣ�����������Ϊ______________������ţ�дһ�鼴�ɣ����������ѡ�������ʽ̼��ͭ����ɣ�д��������̡�____________________________��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�

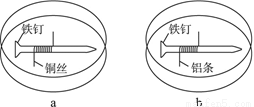
 ������Ԥ����Ѫ�ܼ��������ã���һ�ֺϳ�·�����£�
������Ԥ����Ѫ�ܼ��������ã���һ�ֺϳ�·�����£�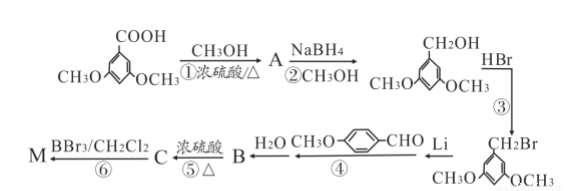
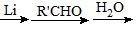


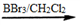
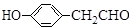
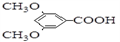 �˴Ź��������в�ͬ��ѧ��������ԭ�Ӹ�����Ϊ___________��
�˴Ź��������в�ͬ��ѧ��������ԭ�Ӹ�����Ϊ___________�� �ж���ͬ���칹�壬�����ܷ���������Ӧ�ұ�����һ��ȡ����ֻ�����ֵ�ͬ���칹����_________________�֡�
�ж���ͬ���칹�壬�����ܷ���������Ӧ�ұ�����һ��ȡ����ֻ�����ֵ�ͬ���칹����_________________�֡� 2SO3(g)��ƽ�ⳣ����ֵΪ_______________��
2SO3(g)��ƽ�ⳣ����ֵΪ_______________��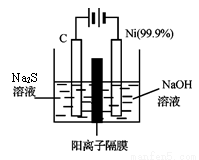
 CO(g) + H2(g) ��H>0����Ҫ��߷�Ӧ������Ҫ���H2O��ת���ʣ����д�ʩ�п��е��ǣ� ��
CO(g) + H2(g) ��H>0����Ҫ��߷�Ӧ������Ҫ���H2O��ת���ʣ����д�ʩ�п��е��ǣ� ��