��Ŀ����
��1���������ʵ���Ũ��Ϊa mol/L�ı�����ȥ�ⶨV mL NaOH��Һ�����ʵ���Ũ�ȣ�����д���пհף���ʽ�ζ���������ˮϴ����Ӧ�ý��еIJ�����______��ͼ1����ʽ�ζ�����Һ���ڵζ�ǰ��Ķ�������c ��NaOH��=______�����ڵζ�ǰ�ζ��ܼ��첿���������ݣ��ζ���ζ��ܼ��첿��������ʧ����ⶨ��NaOH���ʵ���Ũ�Ȼ�______��ƫ�ߡ�ƫ�ͣ���
��2��ͼ2���������������Ƶĵζ�����a��b���������������գ�
����a����______��Һ�ζ�______��Һ������b����______��Һ�ζ�______��Һ����������ʵ���Ũ��Ϊ______mol/L��

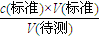 �����жϣ�
�����жϣ���2������a��pH���ߣ��ҿ�ʼpH=1����Ϊ��ζ������b��pH���ͣ��ҿ�ʼpH=13����Ϊ��ζ����pH=1�����������Ũ�ȣ�
����⣺��1���ζ���ʹ��ǰӦ��������ϴ���ʴ�Ϊ���ñ�������ϴ2-3�Σ��к͵ζ��������NaOH�����ʵ�����ȣ���V2-V1��a=c ��NaOH��V���ζ�ǰ��ʽ�ζ��ܼ��첿�������ݣ��ζ�����첿�ֳ�����Һ���ᵼ���������ƫ�������ƫ�ʴ�Ϊ���ñ�������ϴ2-3�Σ�
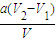 ��ƫ�ߣ�
��ƫ�ߣ���2����ʼpH=1����pH=-lgc��H+������c��HCl��=0.1mol/L������a��pH���ߣ���pH=1����ΪNaOH�ζ�HCl������b��pH���ͣ��ҿ�ʼpH=13����ΪHCl�ζ�NaOH��
�ʴ�Ϊ��NaOH��HCl��HCl��NaOH��0.1��
���������⿼������к͵ζ�ʵ�飬��ȷʵ��ԭ�������衢�����Լ�ע������ɽ��ע�����ͼ��������к͵Ĺ�ϵ��


��1��������������������Եķ���
��2��������ͼ1��ʾװ�ÿ�����ȡ���Ӧ��״����������Ӧ�Ƿ���Ҫ��������
��3��ijͬѧ�����ͼ2��ʾװ�ã��ô�������16.9%ϡ���ᷴӦ��ȡNO���岢̽�����������ļ�̬����ش��й����⣮��֪16.9%ϡ������ܶ�Ϊ1.10g/cm3���������ʵ���Ũ��Ϊ
��4������ֹˮ��a���ر�ֹˮ��bʱ��Aװ�õĸ�����й۲쵽��������
��5����֪���з�Ӧ���Է�����Fe2O3+3KNO3+4KOH
| ���� |
a��HNO3�������Ա�KNO3��
b����Ӧ���¶Ȳ���
c��HNO3�����ȶ��Ա�KNO3��
d��FeO42-���ܴ�����������Һ��
��6������������ҩƷ���Թܺͽ�ͷ�ιܣ�0.1mol/L KSCN��Һ��0.2mol/L����KMnO4��Һ��0.1mol/L KI��Һ����ˮ�ȣ��������һ����ʵ�飬̽��Aװ���ձ�����ȫ��Ӧ�������ܵļ�̬����д����ʵ�鱨�棺
| ʵ�鲽�� | ���� | ��������� |
| ��һ�� | ȡ����Һ��װ���Թܣ����� ���е��뼸��KSCN��Һ�� |
|
| �ڶ��� | ����Һ��ɫ��ȥ����˵������Fe2+�� �������Ա仯����˵������Fe2+�� |
ʵ����� | ��Ʒ������/g | NaOH��Һ�����/mL | ���������/L |
�� | 7.4 | 40.00 | 1.68 |
�� | 14.8 | 40.00 | 3.36 |
�� | 22.2 | 40.00 | 1.12 |
�� | 37.0 | 40.00 | 0.00 |
��1��д��ʵ��������йص����ӷ���ʽ��
��2���ɱ�������ֱ���Ʋ��״����3.7 g��Ʒ����ͬ��ʵ��ʱ�����ɰ��������Ϊ��������
��3���Լ���û������(NH4)2SO4��NH4HSO4�����ʵ���֮�ȡ�
��4���������NaOH��Һ�����ʵ���Ũ����Ӧѡ���_________�����ݣ��ɴ����NaOH��Һ�����ʵ���Ũ��Ϊ���٣�
Ϊ�˷��λ�����Ⱦ����β�������ۺ����ã�ij���᳧�ð�ˮ����β���е�SO2����������Һ�м���Ũ���ᣬ����ȡ��Ũ�ȵ�SO2��(NH4)2SO4��NH4HSO4���塣Ϊ�ⶨ����(NH4)2SO4��NH4HSO4�����������ɣ��ֳ�ȡ��Ʒ�ķݣ��ֱ������ͬŨ�ȵ�NaOH��Һ��40.00 mL��������120 �����ң�ʹ����ȫ���ݳ���(NH4)2SO4��NH4HSO4�ֽ��¶Ⱦ�����200 �桳������й�ʵ���������£���״������
| ʵ����� | ��Ʒ������/g | NaOH��Һ�����/mL | ���������/L |
| �� | 7.4 | 40.00 | 1.68 |
| �� | 14.8 | 40.00 | 3.36 |
| �� | 22.2 | 40.00 | 1.12 |
| �� | 37.0 | 40.00 | 0.00 |
��1��д��ʵ��������йص����ӷ���ʽ��
��2���ɱ�������ֱ���Ʋ��״����3.7 g��Ʒ����ͬ��ʵ��ʱ�����ɰ��������Ϊ��������
��3���Լ���û������(NH4)2SO4��NH4HSO4�����ʵ���֮�ȡ�
��4���������NaOH��Һ�����ʵ���Ũ����Ӧѡ���_________�����ݣ��ɴ����NaOH��Һ�����ʵ���Ũ��Ϊ���٣�
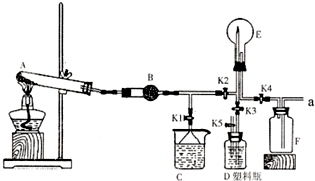 ��ͼ��ʵ������ij��������Ʊ������ʼ��鼰�ռ�װ��ͼ��
��ͼ��ʵ������ij��������Ʊ������ʼ��鼰�ռ�װ��ͼ��