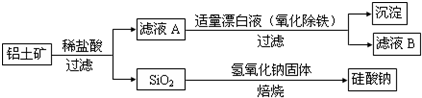
��Ŀ����
8��������A�ķ���ʽΪC9H15OCl�������к���һ����Ԫ����һ����������ֻ��һ��ȡ������F�����в�������A����������֮���ת����ͼ��ʾ��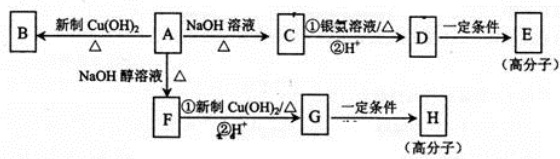
��1��A��F�ķ�Ӧ��������ȥ��Ӧ��G�к��������ŵ�������̼̼˫�����Ȼ���
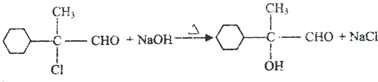
��2��A��C�ķ�Ӧ����ʽ��
 ��
����3��H�Ľṹ��ʽ��
 ��E�Ľṹ��ʽ��
��E�Ľṹ��ʽ�� ��
����4���е�ͬѧ��ΪB�п���û����ԭ�ӣ���Ĺ۵���ͬ�⣨�ͬ�⡱��ͬ�⡱��������������ڼ��Ի�������ԭ���п���ˮ�⣬ʹ��B��û����ԭ�ӣ�
��5��ij���ĺ���������X��������������ͬ���칹���У��˴Ź���������ʾΪ2������HCOOC��CH3��3��д�ṹ��ʽ����ֻ������������ͬ���칹����5�֣�
����Է���������C��54 ����ԭ������C��ͬ ���ܷ���ˮ�ⷴӦ��
���� A�ķ���ʽΪC9H15OCl��A�IJ����Ͷ�=$\frac{9��2+2-16}{2}$=2�������к���һ����Ԫ����һ����������ֻ��һ��ȡ������A������ȥ��Ӧ��F��F�����в�����������AΪ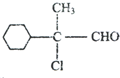 ��A������BΪ
��A������BΪ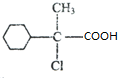 ��A����ˮ���CΪ
��A����ˮ���CΪ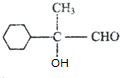 ��C ������DΪ
��C ������DΪ ��D�������۷�Ӧ��EΪ
��D�������۷�Ӧ��EΪ ��A������ȥ��Ӧ��FΪ
��A������ȥ��Ӧ��FΪ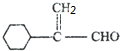 ��F������GΪ
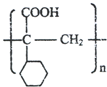
��F������GΪ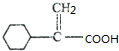 ��G��һ�������·����Ӿ۷�Ӧ��HΪ
��G��һ�������·����Ӿ۷�Ӧ��HΪ ���ݴ˴��⣮
���ݴ˴��⣮
��� �⣺A�ķ���ʽΪC9H15OCl��A�IJ����Ͷ�=$\frac{9��2+2-16}{2}$=2�������к���һ����Ԫ����һ����������ֻ��һ��ȡ������A������ȥ��Ӧ��F��F�����в�����������AΪ ��A������BΪ
��A������BΪ ��A����ˮ���CΪ
��A����ˮ���CΪ ��C ������DΪ
��C ������DΪ ��D�������۷�Ӧ��EΪ
��D�������۷�Ӧ��EΪ ��A������ȥ��Ӧ��FΪ
��A������ȥ��Ӧ��FΪ ��F������GΪ
��F������GΪ ��G��һ�������·����Ӿ۷�Ӧ��HΪ
��G��һ�������·����Ӿ۷�Ӧ��HΪ ��
��
��1����������ķ�����֪��A��F�ķ�Ӧ��������ȥ��Ӧ��GΪ ��G�к��������ŵ�������̼̼˫�����Ȼ���
��G�к��������ŵ�������̼̼˫�����Ȼ���
�ʴ�Ϊ����ȥ��Ӧ��̼̼˫�����Ȼ���
��2��A��C�ķ�Ӧ����ʽ�� ��
��
�ʴ�Ϊ�� ��
��
��3����������ķ�����֪��H�Ľṹ��ʽ�� ��E�Ľṹ��ʽ��
��E�Ľṹ��ʽ�� ��
��
�ʴ�Ϊ�� ��
�� ��
��
��4������ �ڼ��������±�������ͭ������B����������ԭ���ڼ��Ի�������ԭ���п���ˮ�⣬ʹ��B��û����ԭ�ӣ�
�ڼ��������±�������ͭ������B����������ԭ���ڼ��Ի�������ԭ���п���ˮ�⣬ʹ��B��û����ԭ�ӣ�
�ʴ�Ϊ��ͬ�⣻�ڼ��Ի�������ԭ���п���ˮ�⣬ʹ��B��û����ԭ�ӣ�
��Ĺ۵��� ���ͬ�⡱��ͬ�⡱������������ɣ�
��5��CΪ ��ij���ĺ���������X����������������Է���������C��54����X����Է�������Ϊ102������ԭ������C��ͬ��˵����2����ԭ�ӣ����ܷ���ˮ�ⷴӦ��˵�����������˴Ź���������ʾΪ2��壬��XΪHCOOC��CH3��3��ֻ������������ͬ���칹��ΪHCOOCH2CH��CH3��2��HCOOCH��CH3��CH2CH3��CH3COOCH2CH2CH3��CH3CH2COOCH2CH3��CH3CH2CH2COOCH3����5 �֣�
��ij���ĺ���������X����������������Է���������C��54����X����Է�������Ϊ102������ԭ������C��ͬ��˵����2����ԭ�ӣ����ܷ���ˮ�ⷴӦ��˵�����������˴Ź���������ʾΪ2��壬��XΪHCOOC��CH3��3��ֻ������������ͬ���칹��ΪHCOOCH2CH��CH3��2��HCOOCH��CH3��CH2CH3��CH3COOCH2CH2CH3��CH3CH2COOCH2CH3��CH3CH2CH2COOCH3����5 �֣�
�ʴ�Ϊ��HCOOC��CH3��3��5��
���� ���⿼���л����ƶϣ�Ϊ��Ƶ���㣬���ؿ���ѧ�������ƶ���������ȷ�ж�A�ṹ��ʽ�ǽⱾ��ؼ������ݷ�Ӧ�������ijЩ���ʷ���ʽ���ṹ�����ƶϣ��ѵ��ǣ�5��ͬ���칹�������жϣ�Ҫ���ǹ������칹��λ���칹����Ŀ�Ѷ��еȣ�

 �ο�������ϵ�д�
�ο�������ϵ�д� ������ѧ��ʱ��ҵϵ�д�
������ѧ��ʱ��ҵϵ�д� ���������ʱ��ѵϵ�д�
���������ʱ��ѵϵ�д�| ��ѧʽ | CH3COOH | HClO | H2CO3 | H2C2O4 |
Ka | Ka=1.8��10-5 | Ka=3.0��10-8 | Ka1=4.1��10-7 Ka2=5.6��10-11 | Ka1=5.9��10-2 Ka2=6.4��10-5 |
��2��pH ��ͬ��NaClO��CH3COOK��Һ������Һ�����ʵ���Ũ�ȵĴ�С��ϵ�ǣ�CH3COONa��NaClO������Һ�У�[c��Na+��-c��ClO-��]=[c��K+��-c��CH3COO-��]���������������=������
��3���� 0.1mol/LCH3COOH��Һ�еμ�NaOH��Һ��c��CH3COOH����c��CH3COO-��=5��9����ʱ��Һ pH=5��
��4��̼������Һ�еμ�������ˮ�����ӷ���ʽΪCl2+2CO32-+H2O�T2HCO3-+Cl-+ClO-��
| A�� | ��״���£�11.2L���к��е�̼̼˫����ĿΪ1.5NA | |
| B�� | ���³�ѹ�£�2.8g��N2��CO��ɵĻ�������к��е�ԭ����Ϊ0.1NA | |
| C�� | 25��ʱ��1.0LpH=12��Na2CO3��Һ�к��е�������������0.01NA | |
| D�� | �ڷ�Ӧ��Cu2S+O2$\frac{\underline{\;����\;}}{\;}$2Cu+SO2�У�����1molCu��ת�Ƶĵ�����Ϊ2NA |
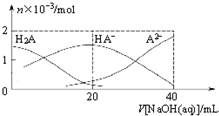 25���£���20mL 0.1mol•L-1H2A��Һ�еμ�0.1mol•L-1NaOH��Һ���й��������ʵ����ı仯��ͼ��ʾ�������й�˵����ȷ���ǣ�������
25���£���20mL 0.1mol•L-1H2A��Һ�еμ�0.1mol•L-1NaOH��Һ���й��������ʵ����ı仯��ͼ��ʾ�������й�˵����ȷ���ǣ�������| A�� | H2A����ǿ�� | |
| B�� | $\frac{c��H{A}^{-}��}{c��{H}_{2}{A}^{\;}��}$����V[NaOH��aq��]���������С | |
| C�� | V[NaOH��aq��]=20mLʱ����Һ�д��ڹ�ϵ��c��HA-��+c��A2-��+c��H2A��=0.1mol•L-1 | |
| D�� | V[NaOH��aq��]=20mLʱ����Һ�д��ڹ�ϵ��c��Na+����c��HA-����c��H+����c��A2-����c��OH-�� |
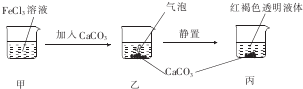
| A�� | �ס�����Һ����ɲ������������ | |
| B�� | ���в�����������ʹBaCl2��Һ����� | |
| C�� | ��������Һ����ı仯�����ձ���c��Cl-������ | |
| D�� | ����CaC03����CaS04��Ҳ�ɵõ���ͬ��ʵ������ |
| A�� | Ϊ�˷�ֹʳƷ�ܳ�����֬ʳƷ�������ʣ����ڰ�װ���з���轺������������������ | |
| B�� | �������к�ʹ���ܱ����オ������;ۺ������ | |
| C�� | ��ɫ��ѧ�ĺ�����Ӧ�û�ѧԭ���Ի�����Ⱦ�������� | |
| D�� | �Ͼɵ��Ӧ���л��գ����������� |
 Na2S2O3�׳ƴ��մ���������Ҫ�Ļ���ԭ�ϣ���Na2SO3�������ˮ��Һ�м��ȷ�Ӧ�������Ƶ�Na2S2O3����֪10���70��ʱ��Na2S2O3��100gˮ�е��ܽ�ȷֱ�Ϊ60��.0g��212g�������£�����Һ�������ľ�����Na2S2O3•5H2O��
Na2S2O3�׳ƴ��մ���������Ҫ�Ļ���ԭ�ϣ���Na2SO3�������ˮ��Һ�м��ȷ�Ӧ�������Ƶ�Na2S2O3����֪10���70��ʱ��Na2S2O3��100gˮ�е��ܽ�ȷֱ�Ϊ60��.0g��212g�������£�����Һ�������ľ�����Na2S2O3•5H2O����ʵ��������ȡNa2S2O3•5H2O���壨Na2S2O3•5H2O�ķ�����Ϊ248���������£�
�ٳ�ȡ12.6g Na2SO3���ձ��У�����80.0mLˮ��
����ȡ4.0g��ۣ��������Ҵ���ʪ�ӵ�������Һ�У�
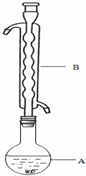
�ۣ���ͼ��ʾ������װ����ȥ����ˮԡ���ȣ��У���ӦԼ1Сʱ����ˣ�
����Һ�ھ�������Ũ������ȴ�ᾧ������Na2S2O3•5H2O���壮
�ݽ��м�ѹ���˲����
��1������B�������������ܻ��������������������������������������Ҵ���ʪ��Ŀ�������ӷ�Ӧ��ĽӴ�������ӿ췴Ӧ���ʣ�
��2����Һ�г�Na2S2O3�Ϳ���δ��Ӧ��ȫ��Na2SO3�⣬����ܴ��ڵ���������Na2SO4�������Һ�и����ʵĺ������ܵͣ�����ķ����ǣ�ȡ��������Һ�����Թܣ���ϡ��������Һ�����Ժ��˵ó�S��������Һ�м�BaCl2��Һ�����а�ɫ��������֤������Na2SO4����֮������
��3��Ϊ�˲��Ʒ�Ĵ��ȣ���ȡ7.40g ��Ʒ�����Ƴ�250mL��Һ������Һ����ȡ25.00mL����ƿ�У��μӵ�����Һ��ָʾ��������Ũ��Ϊ0.0500mol/L �ĵ�ˮ������ʽ�����ʽ����ʽ�����ζ������ζ���2S2O32-+I2=S4O62-+2I-�����ζ�������£�
| �ζ����� | �ζ�ǰ������mL�� | �ζ��ζ��������mL�� |
| ��һ�� | 0.30 | 31.12 |
| �ڶ��� | 0.36 | 31.56 |
| ������ | 1.10 | 31.88 |
| A�� | 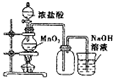 ���MnO2������Ũ����Ϳ�ȫ������ | |
| B�� | 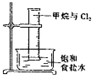 ��Ͳ�з�����ȡ����Ӧ | |
| C�� | 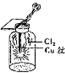 ������ɫ���� | |
| D�� |  �������ɫ������ɫ��˵��������Ư���� |