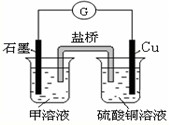
��Ŀ����
A��B��C��D��E����ѧ������5�ֻ����A��B�������Ԫ��X��Y���ĵ����������г����Ľ�����������ʼ�Ĺ�ϵ����ͼ��ʾ��

�ش��������⣺
��1��X�ĵ�����A��Ӧ�Ļ�ѧ����ʽ��______��
��2����C��Һ�Լ��ԣ����Լ�1��______��Һ��д��ѧʽ������C��Һ�����ԣ��������Ե�ԭ���ǣ������ӷ���ʽ��ʾ��______��
��3�����Լ�1���Լ�2����ϡ���ᣮ
�ټ�������D����Һ�н������ӵķ�����______��
��д��D-E��Ӧ�����ӷ���ʽ______��
��ij��Ч��ˮ������Y��OH��SO4�ۺϵõ��ģ���ҵ����E��ϡ�������������Ϊԭ�����Ʊ�Y��OH��SO4����Ӧ����NO���ɣ��÷�Ӧ�Ļ�ѧ����ʽ�ǣ�������ƽ����______��
�⣺Ԫ��X��Y�ĵ����������г����Ľ�����Y�����������XΪAl��YΪFe��A��B���������Al����������Ӧ����Fe����������Al�����������������ᷴӦ������Ӧ����Fe���������������ᷴӦ�����Լ�1ΪNaOH��Һʱ��CΪƫ�����ƣ��Լ�2Ϊ����ʱ��DΪ��������EΪ�����������Լ�1Ϊ����ʱ��CΪ������������Һˮ�������ԣ�
��1��X�ĵ�����A��ӦΪ���ȷ�Ӧ����Al����������Ӧ������������Fe���÷�ӦΪFe2O3+2Al Al2O3+2Fe���ʴ�Ϊ��Fe2O3+2Al
Al2O3+2Fe���ʴ�Ϊ��Fe2O3+2Al Al2O3+2Fe��
Al2O3+2Fe��
��2��CΪ���������Լ�1��ǿ���ΪNaOH��KOH��������ˮ�������ԣ�ˮ�����ӷ�ӦΪAl3++3H2O?Al��OH��3+3H+��
�ʴ�Ϊ��NaOH����KOH����Al3++3H2O?Al��OH��3+3H+��
��3����DΪ�����������������ӵķ���Ϊȡ������Һ���Թ��У��μӼ���KSCN��Һ����Һ���ɫ����֤��ԭ��Һ�к���Fe3+��
�ʴ�Ϊ��ȡ������Һ���Թ��У��μӼ���KSCN��Һ����Һ���ɫ����֤��ԭ��Һ�к���Fe3+��
��D-E��Ӧ�����ӷ���ʽΪ2Fe3++Fe=3Fe2+���ʴ�Ϊ��2Fe3++Fe=3Fe2+��
��EΪ������������ϡ�������������Ϊԭ������Fe��OH��SO4����Ӧ����NO���ɣ��÷�ӦΪ2FeSO4+2NaNO2+H2SO4�T2Fe��OH��SO4+Na2SO4+2NO����
�ʴ�Ϊ��2FeSO4+2NaNO2+H2SO4�T2Fe��OH��SO4+Na2SO4+2NO����
������Ԫ��X��Y�ĵ����������г����Ľ�����Y�����������XΪAl��YΪFe��A��B���������Al����������Ӧ����Fe����������Al�����������������ᷴӦ������Ӧ����Fe���������������ᷴӦ�����Լ�1ΪNaOH��Һʱ��CΪƫ�����ƣ��Լ�2Ϊ����ʱ��DΪ��������EΪ�����������Լ�1Ϊ����ʱ��CΪ������������Һˮ�������ԣ�����ϻ�ѧ��������ɣ�
���������⿼��������ƶϣ���ȷ���ȷ�Ӧ��Al����������Fe�������������ʼ��ɽ�𣬲�ע���Լ�1����Ϊ���ǿ�������������Ŀ�ѶȲ���
��1��X�ĵ�����A��ӦΪ���ȷ�Ӧ����Al����������Ӧ������������Fe���÷�ӦΪFe2O3+2Al
 Al2O3+2Fe���ʴ�Ϊ��Fe2O3+2Al
Al2O3+2Fe���ʴ�Ϊ��Fe2O3+2Al Al2O3+2Fe��
Al2O3+2Fe����2��CΪ���������Լ�1��ǿ���ΪNaOH��KOH��������ˮ�������ԣ�ˮ�����ӷ�ӦΪAl3++3H2O?Al��OH��3+3H+��
�ʴ�Ϊ��NaOH����KOH����Al3++3H2O?Al��OH��3+3H+��
��3����DΪ�����������������ӵķ���Ϊȡ������Һ���Թ��У��μӼ���KSCN��Һ����Һ���ɫ����֤��ԭ��Һ�к���Fe3+��
�ʴ�Ϊ��ȡ������Һ���Թ��У��μӼ���KSCN��Һ����Һ���ɫ����֤��ԭ��Һ�к���Fe3+��
��D-E��Ӧ�����ӷ���ʽΪ2Fe3++Fe=3Fe2+���ʴ�Ϊ��2Fe3++Fe=3Fe2+��
��EΪ������������ϡ�������������Ϊԭ������Fe��OH��SO4����Ӧ����NO���ɣ��÷�ӦΪ2FeSO4+2NaNO2+H2SO4�T2Fe��OH��SO4+Na2SO4+2NO����
�ʴ�Ϊ��2FeSO4+2NaNO2+H2SO4�T2Fe��OH��SO4+Na2SO4+2NO����
������Ԫ��X��Y�ĵ����������г����Ľ�����Y�����������XΪAl��YΪFe��A��B���������Al����������Ӧ����Fe����������Al�����������������ᷴӦ������Ӧ����Fe���������������ᷴӦ�����Լ�1ΪNaOH��Һʱ��CΪƫ�����ƣ��Լ�2Ϊ����ʱ��DΪ��������EΪ�����������Լ�1Ϊ����ʱ��CΪ������������Һˮ�������ԣ�����ϻ�ѧ��������ɣ�
���������⿼��������ƶϣ���ȷ���ȷ�Ӧ��Al����������Fe�������������ʼ��ɽ�𣬲�ע���Լ�1����Ϊ���ǿ�������������Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
��֪A��B��C��D��E�Ƕ�����ԭ�������������������Ԫ�أ�Aԭ����Ԫ�����ڱ���ԭ�Ӱ뾶��С��B��Eͬ���壬��E��ԭ��������B��������C��D�ǽ��������ǵ����������������ˮ������˵������ȷ���ǣ�������
| A�������ӵİ뾶��C��D��E��B | B����ҵ�ϳ��õ�ⷨ�Ƶ�C��D�ĵ��� | C���ȶ��ԣ�A2B��A2E | D������D������ұ��ijЩ���۽��� |
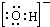
 2DB3����3.2gDB2��ȫת��ΪDB3����ʱ����akJ��1mol DB3������ȫת��Ϊ��ˮ�������bkJ��������33.6L DB2��ȫ�������������Ӧ����
2DB3����3.2gDB2��ȫת��ΪDB3����ʱ����akJ��1mol DB3������ȫת��Ϊ��ˮ�������bkJ��������33.6L DB2��ȫ�������������Ӧ����