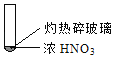��Ŀ����
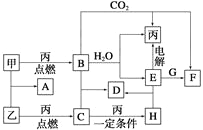
����Ŀ������ý(V2O5)�Ǵ����������õĴ�����Ϊ�ۺ����ã�������Ա����������һ�����ӽ��������շϷ����¹��գ������ʴ�90%���ϡ���֪�Ϸ������к���V2O5��VOSO4�������Բ������������Ͽ�֪��VOSO4������ˮ��V2O5������ˮ��NH4VO3������ˮ���ù��յ���������ͼ��

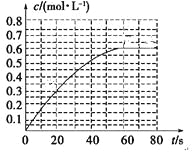
��1��ˮ��ʱ��Ϊ����߷Ϸ��Ľ����ʣ����˷��飬�����Բ�ȡ�Ĵ�ʩ��___________________________________��__________________________________��д��2������
��2��д����Ӧ�ٵ����ӷ���ʽ_______________________________________________��
��3���ù����з�Ӧ�۵ij������ǻ��շ��Ĺؼ��������ʵĸߵͳ�����ҺpHӰ���⣬����Ҫ�����Ȼ��ϵ����NH4Cl������������Һ��V2O5�������ȣ����¶ȡ�������ͼ��������ʵ��Ȼ��ϵ�����¶ȣ��Ȼ��ϵ��Ϊ___________���¶�Ϊ_____________��
��4����Ӧ�ڷ�������Һ�е���Ԫ����Cl������ʽ���ڣ���д����Ӧ�ڵĻ�ѧ����ʽ__________________________________________________________________��
��5���������ϵ�֪��NH4VO3Ҳ��ƫ����泥�����Է�����Ϊ117��20��ʱ��NH4VO3�ܽ��Ϊ0.468g����֪���ӽ�������Һ��c��VO3����=0.10mol/L����Ӧ�۳���ʱ����������0.10mol/L��NH4Cl��Һ��ͨ����ʽ�����жϴ�ʱ�Ƿ���NH4VO3��������������Һ����仯���Բ��ƣ�____________
��6��д�������շ�Ӧ��������NH4VO3�Ʊ�V2O5�Ļ�ѧ����ʽ__________________��
���𰸡� ���� �ʵ������¶ȣ��ӳ�����ʱ�� V2O5+SO32-+4H+=2VO2++SO42-+2H2O 4 80�� 6VOSO4+KClO3+3H2O=3(VO2)2SO4+KCl+3H2SO4 ��20��ʱNH4VO3�ı�����Һ����c(NH4+)=c(VO3-)=0.468��10/117=0.04mol/L��Ksp=1.60��10-3 ,��c(NH4+)��c(VO3-)=(0.10/2)��(0.10/2)=2.50��10-3>Ksp=1.60��10-3 ,���г������� 2NH4VO3 ![]() 2NH3+V2O5+H2O
2NH3+V2O5+H2O
��������(1) ˮ��ʱ��Ϊ����߷Ϸ��Ľ����ʣ����˷��飬�����Բ�ȡ�Ĵ�ʩ���衢�ʵ������¶ȡ��ӳ�����ʱ��ȴ�ʩ��
(2) �����⣬��Ӧ����V2O5����������SO32-����ԭ������Ӧ�����ӷ���ʽΪV2O5+SO32-+4H+=2VO2++SO42-+2H2O��
(3)����ͼ������ɵã�Ҫʹ��������ߣ��ɿ����Ȼ��ϵ��Ϊ4���¶���80�棻
(4) �Ϸ��п���������ΪVOSO4�����Է�Ӧ�ڵĻ�ѧ����ʽΪ6VOSO4+KClO3+ 3H2O= 3(VO2)2SO4+KCl+3H2SO4 ��
(5) �����ܽ�ȵĶ���ɵã���20��ʱNH4VO3�ı�����Һ����c(NH4+)=c(VO3-)= ![]() 0.04mol/L����Ksp(NH4VO3)= (0.04mol/L)2=1.60��10-3 mol2/L2������ʱ��Һ��KQ=c(NH4+)��c(VO3-)=(0.10/2)��(0.10/2)=2.50��10-3 >Ksp(NH4VO3)=1.60��10-3�����г���������
0.04mol/L����Ksp(NH4VO3)= (0.04mol/L)2=1.60��10-3 mol2/L2������ʱ��Һ��KQ=c(NH4+)��c(VO3-)=(0.10/2)��(0.10/2)=2.50��10-3 >Ksp(NH4VO3)=1.60��10-3�����г���������
(6) �����շ�Ӧ��������NH4VO3�Ʊ�V2O5�Ļ�ѧ����ʽΪ2NH4VO3 ![]() 2NH3+ V2O5+ H2O��
2NH3+ V2O5+ H2O��

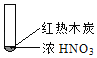
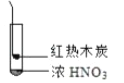
����Ŀ������ʵ���о��к���ɫ����������Աȷ������ý��۲���ȷ���ǣ�������
|
|
|
�� | �� | �� |
A. �����еĺ���ɫ���壬�ƶϲ���������һ���ǻ������
B. ����ɫ���岻�ܱ�������ľ̿��Ũ��������˷�Ӧ
C. ����˵��Ũ������лӷ��ԣ����ɵĺ���ɫ����Ϊ��ԭ����
D. ������������м���CO2���ɴ�˵��ľ̿һ����Ũ���ᷢ���˷�Ӧ