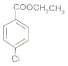
��Ŀ����
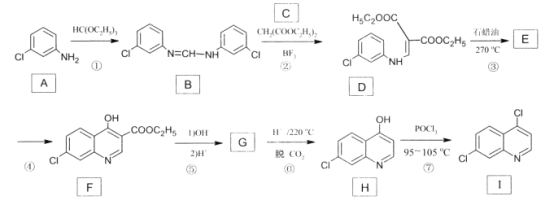
����Ŀ����������ֲ��ϸ���а���ά��ľ����Ϊԭ�Ϻϳ���ʹҩƷĪɳ����(G)��·�ߣ�

��֪��������ૣ�![]() ��������������Ӧ�Ĵ�����
��������������Ӧ�Ĵ�����
��
��
�ش��������⣺
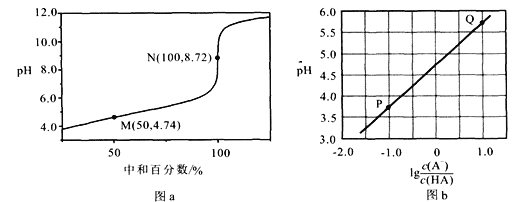
��1��B�й����ŵ�����Ϊ______��
��2��E��G�ķ�Ӧ���ͣ�______��
��3��D��F�Ľṹ��ʽ�ֱ���______��______��
��4��C������������ͭ��Ӧ�Ļ�ѧ����ʽΪ��______��
��5��д��ͬʱ��������������E��һ��ͬ���칹��Ľṹ��ʽ��______��
�ٷ����к��б������ܷ���������Ӧ������FeCl3��Һ������ɫ��Ӧ
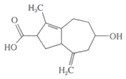
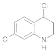
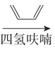
��6���л���H(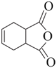 )�Ǻϳɿ�����ҩ�ﰢ����Τ���м��塣�������ϳ�·�ߣ�д����1��3-����ϩΪԭ���Ʊ�
)�Ǻϳɿ�����ҩ�ﰢ����Τ���м��塣�������ϳ�·�ߣ�д����1��3-����ϩΪԭ���Ʊ�![]() �ĺϳ�·������ͼ______�����Լ���ѡ����
�ĺϳ�·������ͼ______�����Լ���ѡ����
![]()
���𰸡��ǻ���ȩ�� ȡ����Ӧ ![]() CH3NHCOOH
CH3NHCOOH 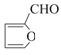 +2Cu(OH)2+NaOH
+2Cu(OH)2+NaOH![]()
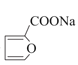 +Cu2O��+3H2O
+Cu2O��+3H2O  (��
(�� ��)
��) 
![]()
![]()
![]()
��������
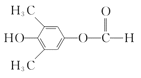
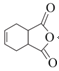
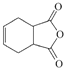
(1)����B�Ľṹ��ʽ��֪B�й�����Ϊȩ�����ǻ���
(2)�Ա�E��G�Ľṹ��֪��E���ǻ���Hԭ�ӱ�-CONHCH3ȡ������G��
(3)��˫ϩ�ϳɷ�Ӧ�����E�Ľṹ��ʽ������E+F��GΪȡ����Ӧ������E��G�ĵĽṹ��ʽ������
(4)C��ȩ����������ͭ����Ϊ-COOH��ͬʱ����������ͭ��ˮ��
(5)E��һ��ͬ���칹�庬�б�����������FeCl3��Һ������ɫ��Ӧ��˵�����з��ǻ����ܷ���������Ӧ�����м����γɵ�����(-OOCH)��
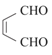
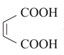
(6)HOCH2CH=CHCH2OH����������OHC-CH=CH-CHO����һ����������![]() ��Ȼ����ȡ����Ӧ����
��Ȼ����ȡ����Ӧ����![]() �������
�������![]() ��������
�������� ��
��
(1)����B�Ľṹ��ʽ��֪B�к���������Ϊȩ�����ǻ���
(2)�Ա�E��G�Ľṹ��֪��E���ǻ���Hԭ�ӱ�CONHCH3ȡ������G������ȡ����Ӧ��
(3)����Ϣ��˫ϩ�ϳɷ�Ӧ�����E�Ľṹ��ʽ��֪DΪ![]() �� E+F��GΪȡ����Ӧ������E��G�ĵĽṹ��ʽ��F�Ľṹ��ʽ����ΪCH3NHCOOH��
�� E+F��GΪȡ����Ӧ������E��G�ĵĽṹ��ʽ��F�Ľṹ��ʽ����ΪCH3NHCOOH��
(4)C��ȩ����������ͭ����ΪCOOH��ͬʱ����������ͭ��ˮ����Ӧ����ʽΪ�� +2Cu(OH)2+NaOH
+2Cu(OH)2+NaOH![]()
 +Cu2O��+3H2O��
+Cu2O��+3H2O��
(5)E��һ��ͬ���칹�庬�б�����������FeCl3��Һ������ɫ��Ӧ��˵�����з��ǻ����ܷ���������Ӧ�����м����γɵ�����(OOCH)����������E��һ��ͬ���칹��ļ�ʽΪ��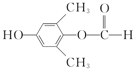 (��
(��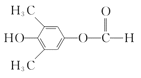 ��)��
��)��
(6) HOCH2CH=CHCH2OH����������OHC-CH=CH-CHO����һ����������![]() ��Ȼ����ȡ����Ӧ����
��Ȼ����ȡ����Ӧ����![]() �������
�������![]() ��������
�������� ����ȫ�ϳ�·������ͼ��
����ȫ�ϳ�·������ͼ��
![]()

![]()
![]()

 ��
��