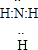��Ŀ����
����������ȷ����
A�������ܱ������н��еķ�Ӧ3A(g) B(g)��C(g)��������������������£��ٳ���һ������A���壬A��ת���ʽ�����
B(g)��C(g)��������������������£��ٳ���һ������A���壬A��ת���ʽ�����
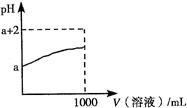
B�����ڿ��淴ӦN2(g)+3H2(g) 2NH3(g)��������Ũ�ȿ����ӻ���Ӱٷ������Ӷ�ʹ��Ӧ�������ӡ�
2NH3(g)��������Ũ�ȿ����ӻ���Ӱٷ������Ӷ�ʹ��Ӧ�������ӡ�
C����AlCl3��Һ��NaAlO2��Һ�ֱ����ɺ����գ����ù�������ΪAl2O3��
D����ӦNH3(g)��HCl(g)��NH4Cl(s) ��H��0 ���κ������¾����Է�����
���𰸡�
A
��������

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
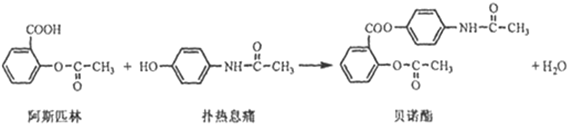
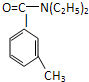 ���ð�������DEET����һ�ֶ��˰�ȫ�����Ը�����ҩ�Ե��������ü�����ṹ��ʽΪ����֪��RCOOH
���ð�������DEET����һ�ֶ��˰�ȫ�����Ը�����ҩ�Ե��������ü�����ṹ��ʽΪ����֪��RCOOH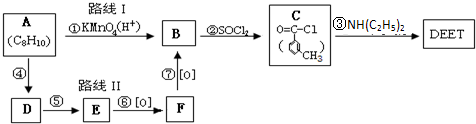


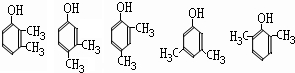 ����д2�֣�
����д2�֣�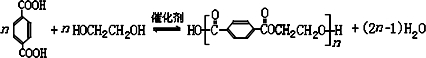

 NH3?H2O+H+
NH3?H2O+H+ ��2010?��������ģ��X��Y��Z��W��Ϊ����10���ӵ���������X��Y��ZΪ���ӣ�WΪ���ӣ���X��Z�����к��еĹ��õ��Ӷ���֮��Ϊ3��4��
��2010?��������ģ��X��Y��Z��W��Ϊ����10���ӵ���������X��Y��ZΪ���ӣ�WΪ���ӣ���X��Z�����к��еĹ��õ��Ӷ���֮��Ϊ3��4��