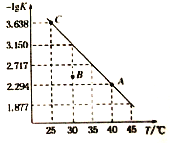
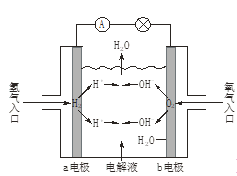
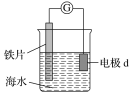
��Ŀ����
����Ŀ��ijͬѧ��ij�ִ��Σ������ܵ�Ca2+��Mg2+�������ᴿʵ�飬�������ͼ��
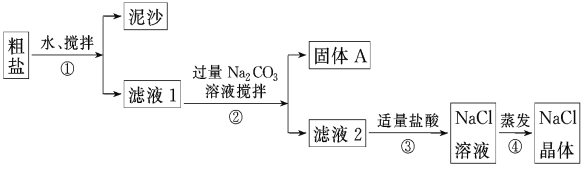
��ش�
��1������ٺ͢ڵIJ���������______________��
��2��������жϼ����������������ķ�����__________________________������ܼ�������ʱҪ�ò��������Ͻ��裬����Ϊ�˷�ֹ________________�������������н϶����������ʱ��Ӧ________________��������ʹˮ�����ɣ�
��3��������A�к�CaCO3��MgCO3��ȡ��������A���Թ��У��μ�ϡ���ᣬ����Ϳ�г���ʯ��ˮ��С�ձ������Թܿ���С�ձ��й۲��������________________________��д��������Ӧ�����ӷ���ʽ_______________________________.
���𰸡����˵μ������������ݷų�Ϊֹ�ֲ����ȣ���ɹ���(����Һ)�ɽ�ֹͣ����(��Ϩ��ƾ���)����ʯ��ˮ�����CO2+ Ca2++ 2OH-��CaCO3��+H2O
��������
��1�������ᴿ����Ϊ���ܽ�����ˡ������ᾧ������ٺ͢ڶ�������˹����Һ�壬���Բ��������ǣ����ˣ���2����Ϊ�����̼���Ʒ�Ӧ�������壬����������Һ�еμ�ϡ���ῴ�������ݲ��������ж�ϡ�������������ܼ�������ʱҪ�ò��������Ͻ��裬����Ϊ�˷�ֹ�ֲ����ȣ����Һ�ηɽ��������������н϶����������ʱ��Ӧֹͣ���Ȼ�Ϩ��ƾ��ƣ�������ʹˮ�����ɣ���3��������A�к�CaCO3��MgCO3��ȡ��������A���Թ��У��μ�ϡ���ᣬ����Ϳ�г���ʯ��ˮ��С�ձ������Թܿ���С�ձ��й۲�������dz���ʯ��ˮ����ǣ�������̼ʹ����ʯ��ˮ����ǣ�������Ӧ�����ӷ���ʽΪCO2+ Ca2++ 2OH-��CaCO3��+H2O��