��Ŀ����
2����ѧ������ϢϢ��أ��밲Ҫ��ش����������еĻ�ѧ���⣺��1����ҵ������������ˮ�Ҫ�õ���ԭ����ʯ��ʯ��
��2����ҵ�Ͻ�����ͨ��ʯ������ȡƯ�ۣ�Ư�۵���Ч�ɷ��ǣ��ѧʽ��Ca��ClO��2��
��3�������кܴ��Σ������ֱ���ƻ�ũ���ʹ�����ữ���γ������ԭ��֮һ��ȼú�в��������壨�ѧʽ��SO2��
��4���������ƿ����ں������DZˮͧ����Ϊ��������Դ��д�����������������̼�Ļ�ѧ��Ӧ����ʽ2Na2O2+2CO2�T2Na2CO3+O2��
��5��С�մ�����������������þ�ȿ�����θ����࣬д��θ��ƽ����Ҫ�ɷ�Ϊ������������θ�ᣨ��Ҫ�ɷ�Ϊϡ���ᣩ���õ����ӷ���ʽAl��OH��3+3H+�TAl3++3H2O��
���� ��1��ʯ��ʯ����Ҫ�ɷ���̼��ƣ�������̼��ƺͶ��������ܷ�����ѧ��Ӧ���ݴ˷�����
��2��Ư��������Ư�����õ���Ca��ClO��2��������ʯ���鷴Ӧ���ɴ�����ơ��Ȼ��ƺ�ˮ��
��3���γ��������Ҫ�ɷ��Ƕ�������
��4���������ƺͶ�����̼��Ӧ����̼���ƺ�������
��5���������������ᷴӦ�����Ȼ�����ˮ��
��� �⣺��1������������ԭ���Ǵ��ʯ��ʯ��ʯӢ������ˮ���������ʯ��ʯΪ��Ҫԭ�ϣ�
�ʴ�Ϊ��ʯ��ʯ��
��2��CaCl2��ǿ��ǿ���β�ˮ�⣬Ca��ClO��2 ��ǿ����������ˮ������HClO��HClO ��Ư���ԣ�����Ư�۵���Ч�ɷ���Ca��ClO��2��
�ʴ�Ϊ��Ca��ClO��2��
��3���γ��������Ҫ�ɷ���SO2��
�ʴ�Ϊ��SO2��
��4���������ƺͶ�����̼��Ӧ����̼���ƺ���������Ӧ����ʽΪ2Na2O2+2CO2�T2Na2CO3+O2��
�ʴ�Ϊ��2Na2O2+2CO2�T2Na2CO3+O2��
��5���������������ᷴӦ�����Ȼ�����ˮ�����ӷ���ʽΪAl��OH��3+3H+�TAl3++3H2O��
�ʴ�Ϊ��Al��OH��3+3H+�TAl3++3H2O��
���� ���⿼��Ԫ�ػ�����֪ʶ���漰���ӷ���ʽ�ͻ�ѧ����ʽ����д��������Ⱦ��֪ʶ�㣬��Ϥ�衢���Ƽ��仯���������Ƶ������ǽ���ؼ�����Ŀ�ѶȲ���

| A�� | 60 g SiO2�����к���Si-O����ĿΪ2NA | |
| B�� | ���³�ѹ�£�1.6 9 O2��O3�Ļ����������������Ϊ0.8NA | |
| C�� | 1 mol/L AICl3��Һ�У�����Al3+��ĿС��NA | |
| D�� | �ܱ�������2 mol NO��l mol 02��ַ�Ӧ������ķ�����Ϊ2NA |
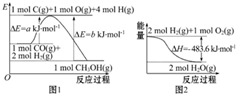
| A�� | ����ͼ1��֪�ϳɼ״����Ȼ�ѧ����ʽΪCO��g��+2H2��g���TCH3OH��g����H1=��b-a��kJ•mol-1 | |
| B�� | ͼ2��ʾ2 mol H2��g�������е�������2 mol��̬ˮ�����е�������483.6 kJ | |
| C�� | 1 mol NaOH�ֱ��1 mol ϡ���ᡢ1 molŨ���ᷴӦ��ǰ�ߵġ�H�Ⱥ��ߵġ�H�� | |
| D�� | ����ȼ��ʱ��ȫ���Ļ�ѧ��ת��Ϊ���� |
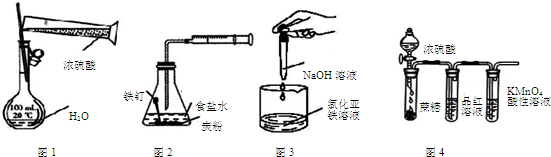
| A�� | ��ͼ1װ������һ�����ʵ���Ũ�ȵ�ϡ���� | |
| B�� | ��ͼ2װ�ÿ�̽������������ʴ | |
| C�� | ��ͼ3װ�ÿ���ȡ���������� | |
| D�� | ��ͼ4װ�ÿ�˵��Ũ���������ˮ�ԡ�ǿ�����ԣ�SO2����Ư���ԡ������� |

| A�� | ����ˮ��CCl4 | B�� | ���� | ||
| C�� | �Ƚ������ε����ȶ��� | D�� | ��ȥCO��CO2 |
| A�� | NaOH��Һ[Ba��OH��2]������Na2SO4��Һ | B�� | HNO3��Һ[HCl]������AgNO3��Һ | ||
| C�� | KOH��Һ[K2CO3]������CaCl2��Һ | D�� | Mg[Al]������NaOH��Һ |
| A�� | AlCl3��Һ�м������ϡ��ˮ��Al3++4NH3•H2O�TAlO2-+4NH4++2H2O | |
| B�� | ��NH4��2Fe��SO4��2��Һ�����NaOH��Һ��Ӧ��Fe��OH��2��Fe2++2OH-�TFe��OH��2�� | |
| C�� | ����ʯ��ˮ�еμ�����NaHCO3��Ca2++2OH++2HCO3-�TCaCO3��+2H2O+CO32- | |
| D�� | ��Mg��HCO3��2��Һ�м�������� NaOH��Һ��Mg2++2HCO3-+4OH-�TMg��OH��2��+2CO32-+2H2O |
 ʵ���������Ƶ�ˮ�������ǽ�I2����KI��Һ�У������Ϳ��Եõ�Ũ�Ƚϴ�ĵ�ˮ����Ҫ����Ϊ�����˷�Ӧ��I2��aq��+I-?I3-������ƽ����ϵ�У�I3-�����ʵ���Ũ��c��I3-�����¶�T�Ĺ�ϵ��ͼ��ʾ�������ϵ�����һ�㶼��ʾƽ��״̬����
ʵ���������Ƶ�ˮ�������ǽ�I2����KI��Һ�У������Ϳ��Եõ�Ũ�Ƚϴ�ĵ�ˮ����Ҫ����Ϊ�����˷�Ӧ��I2��aq��+I-?I3-������ƽ����ϵ�У�I3-�����ʵ���Ũ��c��I3-�����¶�T�Ĺ�ϵ��ͼ��ʾ�������ϵ�����һ�㶼��ʾƽ��״̬����
 ��
��