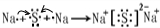��Ŀ����
A��B��C��D�����ֶ�����Ԫ�أ�E�ǹ���Ԫ�أ�A��B��Cͬ���ڣ�C��Dͬ���壬A��ԭ�ӽṹʾ��ͼΪ��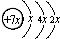 ��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��1������D�Ĺ����ʾʽ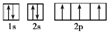

��2���û�ѧʽ��ʾ��������Ԫ��������������Ӧˮ����������ǿ����
��3����Ԫ�ط��ű�ʾD�������ڣ���ϡ������Ԫ���⣩��һ����������Ԫ����
��4��D���⻯���C���⻯��ķе�
��5��EԪ�������ڱ��ĵ�
��6��A��C���������ľ��������Ƿֱ���
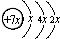 ��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺��1������D�Ĺ����ʾʽ


��2���û�ѧʽ��ʾ��������Ԫ��������������Ӧˮ����������ǿ����
HNO3
HNO3
��������ǿ����NaOH
NaOH
����3����Ԫ�ط��ű�ʾD�������ڣ���ϡ������Ԫ���⣩��һ����������Ԫ����
F
F
����4��D���⻯���C���⻯��ķе�
��
��
����ߡ��͡�����ԭ����������֮�京�����
��������֮�京�����
��D��Һ̬�⻯����һ����Ҫ���ܼ�����������H2O���ƣ�д��B��D��Һ̬�⻯�ﷴӦ�Ļ�ѧ����ʽ2Na+2NH3�T2NaNH2+H2��
2Na+2NH3�T2NaNH2+H2��
����5��EԪ�������ڱ��ĵ�
�������ڵڢ���
�������ڵڢ���
�壬��֪Ԫ�����ڱ��ɰ������Ų���Ϊs����p���ȣ���EԪ����d
d
������6��A��C���������ľ��������Ƿֱ���
ԭ��
ԭ��
���塢����
����
���壮������A��B��C��D�����ֶ�����Ԫ�أ���A��ԭ�ӽṹʾ��ͼ��֪��x=2��A��ԭ������Ϊ14����AΪSiԪ�أ�A��B��Cͬ���ڣ�B��ͬ���ڵ�һ��������С��Ԫ�أ���BΪNaԪ�أ�C��������������ɵ����ӣ���Cԭ�ӵ�3p�ܼ���3�����ӣ���CΪPԪ�أ�C��Dͬ���壬��DΪNԪ�أ�E�ǹ���Ԫ�أ�E����Χ�����Ų�ʽΪ3d64s2��E�ĺ�������Ų�ʽΪ1s22s22p63s23p63d64s2����EΪFeԪ�أ�
����⣺A��B��C��D�����ֶ�����Ԫ�أ���A��ԭ�ӽṹʾ��ͼ��֪��x=2��A��ԭ������Ϊ14����AΪSiԪ�أ�A��B��Cͬ���ڣ�B��ͬ���ڵ�һ��������С��Ԫ�أ���BΪNaԪ�أ�C��������������ɵ����ӣ���Cԭ�ӵ�3p�ܼ���3�����ӣ���CΪPԪ�أ�C��Dͬ���壬��DΪNԪ�أ�E�ǹ���Ԫ�أ�E����Χ�����Ų�ʽΪ3d64s2����EΪFeԪ�أ�
��1��D��NԪ�أ���ԭ�Ӻ�����7�����ӣ����ݹ���ԭ��֪��������ʾʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
��2���ǽ���Խǿ����������Ӧˮ��������Խǿ���ǽ�����N��P��Si��������ǿ����HNO3��������Խǿ����������Ӧˮ�������Խǿ��������Na��ǿ��������ǿ����NaOH��
�ʴ�Ϊ��HNO3��NaOH��
��3��ͬ����������ң���һ��������������⣩�����Ե�һ����������Ԫ����F������������ң��縺�����ʵ縺������Ԫ����F��
�ʴ�Ϊ��F��
��4��D���⻯��ΪNH3����������֮�京��������е��PH3�ߣ�B���ƣ�D���⻯���ǰ������ƺ�Һ���ķ�Ӧ����ʽΪ��2Na+2NH3�T2NaNH2+H2����
�ʴ�Ϊ���ߣ���������֮�京�������2Na+2NH3�T2NaNH2+H2����
��5��EΪFeԪ�أ�E�ĺ�������Ų�ʽΪ1s22s22p63s23p63d64s2���ʺ˵������26��Fe�����ڱ��д��ڵ������ڵڢ��壬�����ڱ��д���d ����
�ʴ�Ϊ���������ڵڢ��壬d��
��6��A��C���������ֱ���SiO2��P2O5���ֱ�����ԭ�Ӿ��塢���Ӿ��壬�ʴ�Ϊ��ԭ�ӣ����ӣ�
��1��D��NԪ�أ���ԭ�Ӻ�����7�����ӣ����ݹ���ԭ��֪��������ʾʽΪ��
 ��
���ʴ�Ϊ��
 ��
����2���ǽ���Խǿ����������Ӧˮ��������Խǿ���ǽ�����N��P��Si��������ǿ����HNO3��������Խǿ����������Ӧˮ�������Խǿ��������Na��ǿ��������ǿ����NaOH��
�ʴ�Ϊ��HNO3��NaOH��
��3��ͬ����������ң���һ��������������⣩�����Ե�һ����������Ԫ����F������������ң��縺�����ʵ縺������Ԫ����F��
�ʴ�Ϊ��F��
��4��D���⻯��ΪNH3����������֮�京��������е��PH3�ߣ�B���ƣ�D���⻯���ǰ������ƺ�Һ���ķ�Ӧ����ʽΪ��2Na+2NH3�T2NaNH2+H2����
�ʴ�Ϊ���ߣ���������֮�京�������2Na+2NH3�T2NaNH2+H2����
��5��EΪFeԪ�أ�E�ĺ�������Ų�ʽΪ1s22s22p63s23p63d64s2���ʺ˵������26��Fe�����ڱ��д��ڵ������ڵڢ��壬�����ڱ��д���d ����
�ʴ�Ϊ���������ڵڢ��壬d��
��6��A��C���������ֱ���SiO2��P2O5���ֱ�����ԭ�Ӿ��塢���Ӿ��壬�ʴ�Ϊ��ԭ�ӣ����ӣ�
���������⿼��Ԫ���ƶϡ�Ԫ�������ɡ���������Ų����ɵȣ��ѶȲ����ƶ�Ԫ���ǽ���Ĺؼ���ע����������Ԫ�����������������ʵĵݱ���ɣ�

��ϰ��ϵ�д�
�����Ŀ
a��b��c��d�����ֶ�����Ԫ�أ�a��b��dͬ���ڣ�c��dͬ���壮a��ԭ�ӽṹʾ��ͼΪ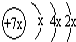 ��b��c�γɻ�����ĵ���ʽΪ
��b��c�γɻ�����ĵ���ʽΪ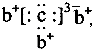 ���бȽ�����ȷ���ǣ�������
���бȽ�����ȷ���ǣ�������
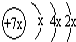 ��b��c�γɻ�����ĵ���ʽΪ
��b��c�γɻ�����ĵ���ʽΪ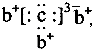 ���бȽ�����ȷ���ǣ�������
���бȽ�����ȷ���ǣ�������| A��ԭ�Ӱ뾶��a��c��d��b | B����ۺ����������c��d��a | C��ԭ��������a��d��b��c | D�����ʵ�������a��b��d��c |
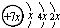 ��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺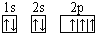

 Al��OH��3+OH-
Al��OH��3+OH-