̀âÄ¿ÄÚÈƯ
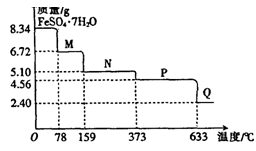
(10·Ö)8£®34gFeS04¡¤7H20ÑùÆ·ÊÜÈÈÍÑË®¹ư³̀µÄÈÈÖØÇúÏß(ÑùÆ·ÖÊÁ¿Ëæζȱ仯µÄÇúÏß)ÈçÏÂͼËùʾ¡£
Çë»Ø´đÏÂÁĐÎỀ⣺
(1)ÊÔÈ·¶¨78¡æʱ¹̀̀åÎïÖÊMµÄ»¯Ñ§Ê½£º ¡£
(2)È¡ÊÊÁ¿380¡æʱËùµĂµÄÑùÆ·P£¬¸ô¾ø¿ƠÆø¼ÓÈÈÖÁ650¡æ£¬µĂµ½̉»ÖÖ¹̀̀åÎïÖÊQ£¬Í¬Ê±ÓĐÁ½ÖÖÎ̃É«Æø̀åÉú³É£¬Đ´³ö¸Ă·´Ó¦µÄ»¯Ñ§·½³̀ʽ
(3)ijĐËÈ¤Đ¡×éÓĂÈçͼËùʾװÖĂÉè¼ÆʵÑ飬ÑéÖ¤(2)ÖĐÉú³ÉµÄÆø̀¬ÎïÖÊ£¬²¢²â¶¨̉Ñ·Ö½âµÄPµÄÖÊÁ¿(²»¿¼ÂÇ×°ÖĂÄÚ¿ƠÆøµÄÓ°Ḯ)¡£
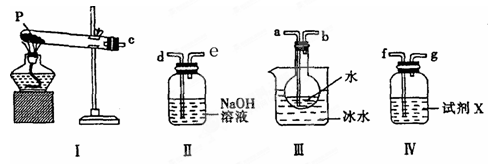
¢ÙÊÔ¼ÁXµÄĂû³ÆÊÇ ¡£
¢Ú°´ÆøÁ÷·½Ị̈Á¬½Ó¸÷̉ÇÆ÷£¬ÓĂ×Öĸ±íʾ½Ó¿ÚµÄÁ¬½Ó˳Đ̣£ºc¡ú ¡£
¢Û³ä·Ö·´Ó¦ºó£¬ÀûÓĂ×°ÖĂIIIÖĐÔ²µ×ÉƠÆ¿ÄÚ»́ºÏÎï²â¶¨̉Ñ·Ö½âµÄPµÄÖÊÁ¿£¬Æä²Ù×÷²½ÖèΪ£ºµÚ̉»²½£ºỊ̈Ô²µ×ÉƠÆ¿ÖĐÖđµÎ¼ÓÈëÂÈ»¯±µÈÜ̉º£¬Ö±ÖÁ³ÁµíÍêÈ«£»µÚ¶₫²½£º¹ưÂË»́ºÏÎÔÚ¹ưÂËÆ÷ÉϽ«³ÁµíÏ´¾»ºó£¬ºæ¸É²¢ÀäÈ´ÖÁỂΣ¬³ÆÖØ¡£µÚÈư²½£º¼̀Đøºæ¸É¡¢ÀäÈ´²¢³ÆÁ¿Ö±ÖÁÁ¬ĐøÁ½´Î³ÆÁ¿µÄÖÊÁ¿²î²»³¬¹ư0£®1gΪֹ¡£Èô×îÖƠµĂµ½³ÁµíµÄÖÊÁ¿ÎªWg£¬Ộ̉Ñ·Ö½âµÄPµÄÖÊÁ¿ (̀î¼ÆËăʽ) ¡£

Çë»Ø´đÏÂÁĐÎỀ⣺
(1)ÊÔÈ·¶¨78¡æʱ¹̀̀åÎïÖÊMµÄ»¯Ñ§Ê½£º ¡£
(2)È¡ÊÊÁ¿380¡æʱËùµĂµÄÑùÆ·P£¬¸ô¾ø¿ƠÆø¼ÓÈÈÖÁ650¡æ£¬µĂµ½̉»ÖÖ¹̀̀åÎïÖÊQ£¬Í¬Ê±ÓĐÁ½ÖÖÎ̃É«Æø̀åÉú³É£¬Đ´³ö¸Ă·´Ó¦µÄ»¯Ñ§·½³̀ʽ
(3)ijĐËÈ¤Đ¡×éÓĂÈçͼËùʾװÖĂÉè¼ÆʵÑ飬ÑéÖ¤(2)ÖĐÉú³ÉµÄÆø̀¬ÎïÖÊ£¬²¢²â¶¨̉Ñ·Ö½âµÄPµÄÖÊÁ¿(²»¿¼ÂÇ×°ÖĂÄÚ¿ƠÆøµÄÓ°Ḯ)¡£

¢ÙÊÔ¼ÁXµÄĂû³ÆÊÇ ¡£
¢Ú°´ÆøÁ÷·½Ị̈Á¬½Ó¸÷̉ÇÆ÷£¬ÓĂ×Öĸ±íʾ½Ó¿ÚµÄÁ¬½Ó˳Đ̣£ºc¡ú ¡£
¢Û³ä·Ö·´Ó¦ºó£¬ÀûÓĂ×°ÖĂIIIÖĐÔ²µ×ÉƠÆ¿ÄÚ»́ºÏÎï²â¶¨̉Ñ·Ö½âµÄPµÄÖÊÁ¿£¬Æä²Ù×÷²½ÖèΪ£ºµÚ̉»²½£ºỊ̈Ô²µ×ÉƠÆ¿ÖĐÖđµÎ¼ÓÈëÂÈ»¯±µÈÜ̉º£¬Ö±ÖÁ³ÁµíÍêÈ«£»µÚ¶₫²½£º¹ưÂË»́ºÏÎÔÚ¹ưÂËÆ÷ÉϽ«³ÁµíÏ´¾»ºó£¬ºæ¸É²¢ÀäÈ´ÖÁỂΣ¬³ÆÖØ¡£µÚÈư²½£º¼̀Đøºæ¸É¡¢ÀäÈ´²¢³ÆÁ¿Ö±ÖÁÁ¬ĐøÁ½´Î³ÆÁ¿µÄÖÊÁ¿²î²»³¬¹ư0£®1gΪֹ¡£Èô×îÖƠµĂµ½³ÁµíµÄÖÊÁ¿ÎªWg£¬Ộ̉Ñ·Ö½âµÄPµÄÖÊÁ¿ (̀î¼ÆËăʽ) ¡£

¢ÅFeS04¡¤7H20
 FeSO4¡¤XH2O£«(7-x)H2O
FeSO4¡¤XH2O£«(7-x)H2O278 18(7¨DX)
8.34 8.34¨D6.72
½âµĂX=4
FeS04¡¤4H20
¢Æ2FeSO4
 Fe2O3£«SO2 ¡ü£«SO3¡ü
Fe2O3£«SO2 ¡ü£«SO3¡ü¢Ç¢ÙÆ·º́ÈÜ̉º¢Úa¡úb¡úf¡úg¡úd
¢È2FeSO4 ~ SO3 ~ BaSO4
2¡Á152 233
m w


Á·Ï°²áϵÁĐ´đ°¸
 ĂûĐ£¿Î̀ĂϵÁĐ´đ°¸
ĂûĐ£¿Î̀ĂϵÁĐ´đ°¸
Ïà¹Ø̀âÄ¿

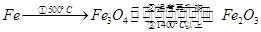
 £»
£» ²¢Ö¸Ă÷¸ĂÑơ»¯»¹Ô·´Ó¦µÄ»¹Ô¼ÁÊÇ £¬Ñơ»¯¼ÁÊÇ ¡£
²¢Ö¸Ă÷¸ĂÑơ»¯»¹Ô·´Ó¦µÄ»¹Ô¼ÁÊÇ £¬Ñơ»¯¼ÁÊÇ ¡£
 Èëµ½CuSO4ÈÜ̉ºÖĐ
Èëµ½CuSO4ÈÜ̉ºÖĐ 4H+ + O2¡ü+ 2Cu
4H+ + O2¡ü+ 2Cu