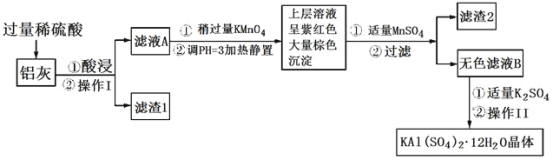
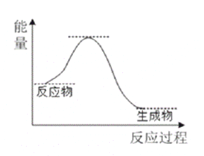
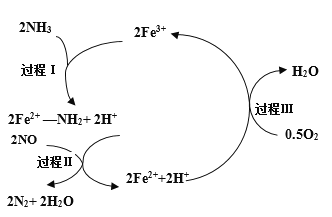
��Ŀ����
����Ŀ��������ʵ���Ӧ�ķ���ʽ��������ǣ� ��
A.��֪��4NH3(g)+5O2(g)=4NO(g)+6H2O(g) ��H<0�����ȵIJ�˿��NH3��O2������Ӵ�����˿�������ֺ���
B.��֪��2NO2(g)������ɫ��![]() N2O4(g)����ɫ�� ��H<0������NO2������������ˮ�У���ɫ��dz
N2O4(g)����ɫ�� ��H<0������NO2������������ˮ�У���ɫ��dz
C.��H+(aq)+OH-(aq)=H2O(l) ��H=-57.3kJ��mol-1������0.1molHCl�������м���4.0gNaOH���壬�ų�����������5.73kJ
D.����ȼ�յ��Ȼ�ѧ����ʽ�ɱ�ʾΪ��CH4(g)+2O2(g)=CO2(g)+2H2O(l) ��H=-890.3kJ��mol-1��������ȼ����Ϊ890.3kJ��mol-1
���𰸡�C
��������
A���ÿ��淴Ӧ���ȣ����ȵIJ�˿���յ���Ӧ�ų��������������ֺ��ȣ�A��ȷ��
B���÷�ӦΪ���ȷ�Ӧ�������¶ȵ�ʱ��ƽ���������ƶ���������ɫ��dz��B��ȷ��
C��NaOH�������������л�Ҫ�ų��������ȣ����Ȼ�ѧ����ʽ������ѡ���еķ���ʽ���C����
D��ȼ��������1mol��������ȫȼ�������ȶ����������ų����������Ȼ�ѧ����ʽ��������Ϊ�ȶ������D��ȷ��
��ѡC��

 �ǻ�С��ϰϵ�д�
�ǻ�С��ϰϵ�д�����Ŀ��T1��ʱ�����ݻ�Ϊ2L���ܱ������г���һ������A(g)��B(g)��������Ӧ��2A(g)+B(g)![]() 2C(g)��һ��ʱ���Ӧ�ﵽƽ��״̬����Ӧ�����вⶨ�IJ����������±���ʾ��
2C(g)��һ��ʱ���Ӧ�ﵽƽ��״̬����Ӧ�����вⶨ�IJ����������±���ʾ��
��Ӧʱ��/min | n(A)/mol | n(B)/mol |
0 | 2 | 1 |
5 | 1.2 | |
10 | 0.4 | |
15 | 0.8 |
����˵��������ǣ���
A.ǰ5min��ƽ����Ӧ����Ϊv(A)=0.08mol��L-1��min-1
B.�����¶�ΪT2��ʱ�����������Ӧ��ƽ�ⳣ��Ϊ20��������ӦΪ���ȷ�Ӧ
C.�����¶Ȳ��䣬��ƽ�����������ٳ���0.2molA(g)��0.2molB(g)ʱ��v(��)>v(��)
D.��ͬ�¶��£�����ʼʱ�������г���0.2molC(g)���ﵽƽ��״̬ʱC��ת����Ϊ40%
����Ŀ��ij�о�С���Ա�����ۼ���Ũ�����ϡ����ķ���ʱ���ռ������·�����
���� | ���� | |
�� | ����ֱ�������� | �����ΪŨ���� |
�� | �ֱ���������Ƭ | �����̼�����ζ��ΪŨ���� |
�� | �ֱ����ʢˮ���Թ��� | ������ΪŨ���� |
�� | �ֱ��� | �����ΪŨ���� |
(1)������______
(2)������һ�����������Ľ����ܳ�Ϊ���з�������______���Ľ�������______��
(3)������������ּ�������ɱ���
���� | ���� | |
A | __________ | ___________ |
B | ___________ | __________ |