��Ŀ����
ij��Һ�п��ܺ�������6�������еļ��֣�NH4+��A13+��Mg2+��CO32�D��Clһ��SO42�D��Ϊȷ����Һ��ɣ���ȡ100 mL�ֳ����ȷ���Һ��������ʵ�飺
��1�����һ����Һ�м��� AgNO3��Һ�г���������
��2����ڶ�����Һ�м�������NaOH��Һ��ַ�Ӧ�����յõ�����0.58 g(�����ˡ�ϴ�ӡ������ͬ)��ͬʱ�ռ�������0.03 mol(������ȫ������Һ���ݳ�)��
��3�����������Һ�м�������BaCl2��Һ(�����ữ)��ַ�Ӧ�õ�����6.99 g��
�ɴ˿�֪�����й���ԭ��Һ��ɵ���ȷ������
��1�����һ����Һ�м��� AgNO3��Һ�г���������
��2����ڶ�����Һ�м�������NaOH��Һ��ַ�Ӧ�����յõ�����0.58 g(�����ˡ�ϴ�ӡ������ͬ)��ͬʱ�ռ�������0.03 mol(������ȫ������Һ���ݳ�)��
��3�����������Һ�м�������BaCl2��Һ(�����ữ)��ַ�Ӧ�õ�����6.99 g��
�ɴ˿�֪�����й���ԭ��Һ��ɵ���ȷ������
| A��һ������Clһ |
| B��һ������A13+��NH4+ |
| C��һ��������Mg2+�����ܴ���A13+ |
| D����Һ��SO42�D��Ũ����0. 3 mol/L |
B
������������� AgNO3��Һ�г�����������ȷ����Һ���Ƿ�һ������Clһ��Ϊ̼����Ҳ�ǰ�ɫ������A����������þ�������������ƣ����Լ���������������Ʋ�������˵����Һ��һ������Mg2+����Һ�м�������BaCl2��Һ(�����ữ)��ַ�Ӧ�õ����ᱵ����6.99 g�����ʵ���
 ������Ԫ���غ���������ӵ����ʵ���Ϊ
������Ԫ���غ���������ӵ����ʵ���Ϊ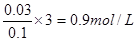 ��D�������Դ�ѡB��
��D�������Դ�ѡB��
��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
 ��MgCl2��CuCl2�����Һ����μ��백ˮ,��������ɫ����,˵��Ksp[Cu(OH)2]>Ksp[Mg(OH)2]
��MgCl2��CuCl2�����Һ����μ��백ˮ,��������ɫ����,˵��Ksp[Cu(OH)2]>Ksp[Mg(OH)2] H+ + A2����������˵����ȷ����( )
H+ + A2����������˵����ȷ����( ) H+ +A2����
H+ +A2����