��Ŀ����
15����1���ϳɰ���ҵ�ĺ��ķ�Ӧ��N2��g��+3H2��g���T2NH3��g����H=Q kJ•mol-1����Ӧ�����������仯��ͼ1��ʾ���ش��������⣮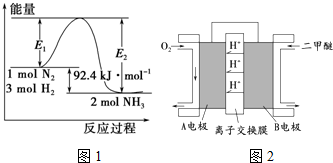
���ڷ�Ӧ��ϵ�м����������Ӧ��������E1�ı仯��E1��С�������������С�����䡱����
����500�桢2��107 Pa�ʹ�����������һ�ܱ������г���0.5mol N2��1.5mol H2����ַ�Ӧ�ų��������������������������=����46.2kJ��
�۹��ڸ÷�Ӧ������˵���У���ȷ����C������ĸ����
A����H��0����S��0 B����H��0����S��0
C����H��0����S��0D����H��0����S��0
��2����ͼ2�Ƕ����ѣ�CH3OCH3��ȼ�ϵ�صĹ���ԭ��ʾ��ͼ����ش��������⣺
��B�缫�ϵĵ缫��Ӧʽ��CH3OCH3+3H2O-12e-�T2CO2+12H+��
����ȼ�ϵ����ͨ������ѣ��е�Ϊ-24.9�棩������Ϊ2.24L•min-1����״�������Ը�ȼ�ϵ����Ϊ��Դ�����2mol•L-1 CuCl2��Һ500mL�����缫��Ϊ���Ե缫������ͨ��30s������������������15.98g �������������������У�����������Ϊ75%����������С�������λ���֣�
���� ��1���ټ�����������ͷ�Ӧ�Ļ�ܣ�
�ڸ��ݿ��淴Ӧ���ܷ�Ӧ�����ص������
�����ݷ�Ӧ������������Ӧ���ȷ�Ӧ�ʱ�С��0���ر�С��0��
��2��������ͼʾ����ͨ��������һ��Ϊ������ͨ������ѵ�һ��Ϊ�������������ҺΪ���Ի�����������ʧ�������ɶ�����̼�����ݵ����غ�д���缫��Ӧ��
�����ݶ�����ͨ������������ʵ�������ϵ����غ��������������������
��� �⣺��1���ټ�����������ͷ�Ӧ�Ļ�ܣ���E1��E2����С�������֮��䣬��Ӧ�Ȳ��䣬�ʴ�Ϊ����С��
�ڵ�0.5mol N2��1.5mol H2��ȫ��Ӧʱ�����ܷų�46.2kJ������������ӦΪ���ܷ�Ӧ��������ȫ���У����ܱ������г���0.5mol N2��1.5mol H2����ַ�Ӧ�ų�������С��46.2kJ���ʴ�Ϊ������
��N2��g��+3H2��g���T2NH3��g����H=Q kJ•mol-1����Ӧ�����������С�ķ��ȷ�Ӧ����Ӧ���ʱ��H��0����S��0���ʴ�Ϊ��C��
��2���ٷ�Ӧ�����Ƕ����ѵ�ȼ�գ�ԭ��ظ�������������Ӧ���������ڸ����ŵ磬������Ӧ��ԭ��Ӧ�������������ŵ磮��ͼ��֪��a��Ϊ������bΪ�����������ѷŵ����ɶ�����̼�������ӣ�a�缫�ĵ缫��ӦʽΪ CH3OCH3-12e-+3H2O�T2CO2+12H+����ȼ�ϵ�ص�����ϰ���������Ӧ����ȫ������̼����ͨ��缫������ͨ����
�ʴ�Ϊ��CH3OCH3+3H2O-12e-�T2CO2+12H+��
������2.24L•min-1����״�������Ը�ȼ�ϵ����Ϊ��Դ�����2mol•L-1 CuCl2��Һ500mL�����缫��Ϊ���Ե缫������ͨ��30s��ͨ����������ʵ���=$\frac{2.24L/mol��0.50min}{22.4L/mol}$=0.05mol��
���ݵ缫��Ӧ�����غ㣬CH3OCH3--12e---6Cl2�����������У�����������Ϊ75%��
n��Cl2��=0.05mol��75%��6=0.225mol
m��Cl2��=0.225mol��71g/mol=15.98g
�ʴ�Ϊ��15.98��
���� ���⿼���˴����Ի�ܵ�Ӱ�졢���淴Ӧ���ʱ��Լ�ͼ�����Ӧ�ã�ԭ��ص缫��Ӧ����д�����������غ�ļ���Ӧ�ã���Ŀ�Ѷ��еȣ�

 ��У����ϵ�д�
��У����ϵ�д�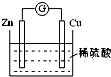
| A�� | ZnƬ������ | B�� | H+��ͭƬ�ϵõ��� | ||
| C�� | ��Ӧ�����е���ת��Ϊ��ѧ�� | D�� | ���Ӵ�пƬͨ��������Һ����ͭƬ |
| A�� | һ�������£�0.1molSO2������������Ӧ����SO3��ת�Ƶ�����Ϊ0.2NA | |
| B�� | ��״���£�20gD2O����������������Ϊ10NA | |
| C�� | ��״���£�22.4LCl2��ȫ����ˮʱ�γ���ˮʱ��ת�Ƶ�����ΪNA | |
| D�� | ����ȼ�ϵ����������22.4L����ʱ����·��ͨ���ĵ�����ĿΪ4NA |
| A�� | FeSO4������Һ��¶�ڿ����У�4Fe2++O2+4H+�T4Fe3++2H2O | |
| B�� | ����ʯ��ˮ��̼����Ʒ�Ӧ��Ca2++2HCO3-+2OH-�TCaCO3��+CO32-+2H2O | |
| C�� | ���Ȼ�����Һ�еμӹ�����ˮ��Al3++4NH3•H2O�TAlO2-+4NH4++2H2O | |
| D�� | H2SO4��Ba��OH��2��Һ��Ӧ��Ba2++OH-+H++SO42-�TBaSO4��+H2O |
| A�� | 15 g | B�� | 22.4 g | C�� | 28 g | D�� | ������ |
| A�� | M��ȫ������N��Ϊ4s2��ԭ�Ӻͺ�������Ų�Ϊ1s22s22p63s23p63d64s2 ��ԭ�� | |
| B�� | 2p�ܼ���һ��δ�ɶԵ��ӵĻ�̬ԭ�Ӻ�ԭ�ӵļ۵����Ų�Ϊ2s22p5 | |
| C�� | 3p�ܼ���һ���չ���Ļ�̬ԭ�Ӻͺ�����ӵ��Ų�Ϊ1s22s22p63s23p2 | |
| D�� | �����������Ǻ������������1/5��ԭ�Ӻͼ۵����Ų�Ϊ4s24p1��ԭ�� |
| A�� | �����Ȼ�����Һ��Ӧ��Fe+Fe3+�T2Fe2+ | |
| B�� | ����ˮ��Ӧ��Na+H2O�TNa++OH-+H2�� | |
| C�� | ϡ����������������Һ��Ӧ��H++OH-�TH2O | |
| D�� | ��������������Һ��Ӧ��2Al+2OH-+6H2O�T2[Al��OH��4]-+3H2�� |
| A�� | CaCO3+2HCl�TCaCl2+H2O+CO2�� | B�� | BaCl2+Na2SO4�T2NaCl+BaSO4�� | ||
| C�� | 2NaOH+CO2�TNa2CO3+H2O | D�� | Zn+CuSO4�TZnSO4+Cu |
| A�� | ���۵������ǦҼ�������˫������һ���Ҽ���һ���м� | |
| B�� | �Ҽ������Ƽ�����ת���м�һ�������Ƽ�����ת | |
| C�� | H2 �����еĦҼ���s-s �Ҽ���HClO�����еĦҼ���p-p �Ҽ� | |
| D�� | C2H4��������5���Ҽ���һ���м���N2H4��ֻ��5���Ҽ� |