��Ŀ����
�����������ض�����Ҫ�Ĺ�ҵ��Ʒ����ش�:
(1)��ҵұ�����Ļ�ѧ����ʽ������������������������
(2)��������������Һ��Ӧ�����ӷ���ʽ��������������������������������
(3)��ҵƷ�������ص���Һ�к���ijЩ����������,�������ӽ���Ĥ������ᴿ��
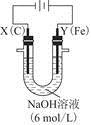
������װ�������ӽ���Ĥ(ֻ����������ͨ��),�乤��ԭ����ͼ��ʾ��
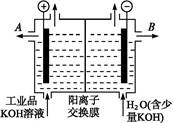
�ٸõ��۵�������Ӧʽ������������������������
��ͨ�翪ʼ��,����������ҺpH������,�����ԭ��������������������������
�۳�ȥ���ʺ������������Һ����Һ������������(��д��A����B��)������
(1)��ҵұ�����Ļ�ѧ����ʽ������������������������
(2)��������������Һ��Ӧ�����ӷ���ʽ��������������������������������
(3)��ҵƷ�������ص���Һ�к���ijЩ����������,�������ӽ���Ĥ������ᴿ��
������װ�������ӽ���Ĥ(ֻ����������ͨ��),�乤��ԭ����ͼ��ʾ��

�ٸõ��۵�������Ӧʽ������������������������
��ͨ�翪ʼ��,����������ҺpH������,�����ԭ��������������������������
�۳�ȥ���ʺ������������Һ����Һ������������(��д��A����B��)������
(1)2Al2O3 4Al+3O2��
4Al+3O2��
(2)2Al+2OH-+2H2O 2Al+3H2��
2Al+3H2��
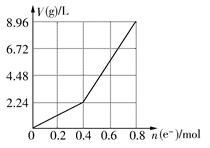
(3)��4OH--4e- 2H2O+O2������H+�ŵ�,�ٽ�ˮ�ĵ���,OH-Ũ������B
2H2O+O2������H+�ŵ�,�ٽ�ˮ�ĵ���,OH-Ũ������B
 4Al+3O2��
4Al+3O2��(2)2Al+2OH-+2H2O
 2Al+3H2��
2Al+3H2��(3)��4OH--4e-
 2H2O+O2������H+�ŵ�,�ٽ�ˮ�ĵ���,OH-Ũ������B
2H2O+O2������H+�ŵ�,�ٽ�ˮ�ĵ���,OH-Ũ������B���ӽ���Ĥ������ᴿKOHʱ,������ӦΪ:4OH--4e- 2H2O+O2��,������ӦΪ:4H2O+4e-
2H2O+O2��,������ӦΪ:4H2O+4e- 4OH-+2H2����������������������K+ͨ�������ӽ���Ĥ�������ƶ�,����������Ӳ���ͨ�����ӽ���Ĥ��һ��ʱ���,��������K+��OH-Ũ�ȱ��,��B��(������)�õ���Һ,�����ᾧ��ɵýϴ�����KOH��
4OH-+2H2����������������������K+ͨ�������ӽ���Ĥ�������ƶ�,����������Ӳ���ͨ�����ӽ���Ĥ��һ��ʱ���,��������K+��OH-Ũ�ȱ��,��B��(������)�õ���Һ,�����ᾧ��ɵýϴ�����KOH��
 2H2O+O2��,������ӦΪ:4H2O+4e-
2H2O+O2��,������ӦΪ:4H2O+4e- 4OH-+2H2����������������������K+ͨ�������ӽ���Ĥ�������ƶ�,����������Ӳ���ͨ�����ӽ���Ĥ��һ��ʱ���,��������K+��OH-Ũ�ȱ��,��B��(������)�õ���Һ,�����ᾧ��ɵýϴ�����KOH��
4OH-+2H2����������������������K+ͨ�������ӽ���Ĥ�������ƶ�,����������Ӳ���ͨ�����ӽ���Ĥ��һ��ʱ���,��������K+��OH-Ũ�ȱ��,��B��(������)�õ���Һ,�����ᾧ��ɵýϴ�����KOH��
��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
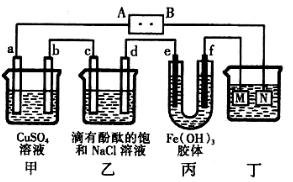

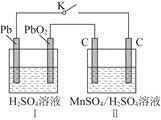
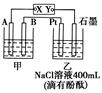
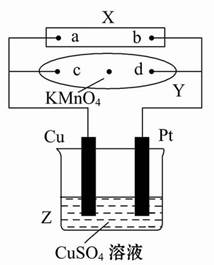
 ��ʵ������У������������������Y������Һ������
��ʵ������У������������������Y������Һ������