��Ŀ����
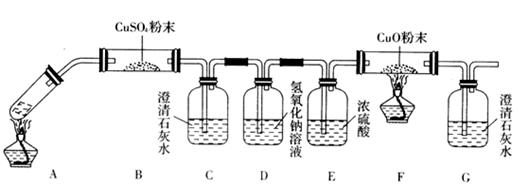
��16�֣�����������Ϣ��ҵ����Ҫ�Ļ������ϡ�ͨ����̼�ڸ����»�ԭ���������Ƶôֹ裨�����������������ʣ����ֹ���������Ӧ�������Ȼ��裨��Ӧ�¶�450-500��C�������Ȼ��辭�ᴿ����������ԭ�ɵøߴ��衣������ʵ�����Ʊ����Ȼ����װ��ʾ��ͼ��


 �����Ϣ���£�
�����Ϣ���£�
 a.���Ȼ�����ˮ����ˮ�⣻
a.���Ȼ�����ˮ����ˮ�⣻
 b.�������������ڸ����¾���������ֱ�ӷ�Ӧ������Ӧ���Ȼ��
b.�������������ڸ����¾���������ֱ�ӷ�Ӧ������Ӧ���Ȼ��
 c.�й����ʵ������������±���
c.�й����ʵ������������±���


 ��ش��������⣺
��ش��������⣺
 ��1��д��װ��A�з�����Ӧ�����ӷ���ʽ ��
��1��д��װ��A�з�����Ӧ�����ӷ���ʽ ��
 ��2��װ��A��g�ܵ������� ��װ��C�е��Լ��� ��װ��E�е�hƿ��Ҫ��ȴ������ ��
��2��װ��A��g�ܵ������� ��װ��C�е��Լ��� ��װ��E�е�hƿ��Ҫ��ȴ������ ��
 ��3��װ��E��hƿ�ռ����Ĵֲ����ͨ���������ƶ�����õ��ߴ������Ȼ��裬�����IJ������У�����Ԫ������ܻ����е�����Ԫ���� ����дԪ�ط��ţ���
��3��װ��E��hƿ�ռ����Ĵֲ����ͨ���������ƶ�����õ��ߴ������Ȼ��裬�����IJ������У�����Ԫ������ܻ����е�����Ԫ���� ����дԪ�ط��ţ���
 ��4��Ϊ�˷�������������Ԫ�صĺ������Ƚ�������Ԥ����������Ԫ�ػ�ԭ��Fe2+������KMnO4����Һ�����������½���������ԭ�ζ�����Ӧ�����ӷ���ʽ�ǣ�
��4��Ϊ�˷�������������Ԫ�صĺ������Ƚ�������Ԥ����������Ԫ�ػ�ԭ��Fe2+������KMnO4����Һ�����������½���������ԭ�ζ�����Ӧ�����ӷ���ʽ�ǣ�

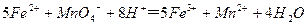
 �ٵζ�ǰ�Ƿ�Ҫ�μ�ָʾ���� ����ǡ�������˵������ ��
�ٵζ�ǰ�Ƿ�Ҫ�μ�ָʾ���� ����ǡ�������˵������ ��
 ��ijͬѧ��ȡ5.000g�����Ԥ������������ƿ�����Ƴ�100ml��Һ����ȡ25.00ml��������Һ����1.000��10-2mol�� L-1KMnO4����Һ�ζ����ﵽ�ζ��յ�ʱ�����ı���Һ20.00ml,�����������Ԫ�ص����������� ��
��ijͬѧ��ȡ5.000g�����Ԥ������������ƿ�����Ƴ�100ml��Һ����ȡ25.00ml��������Һ����1.000��10-2mol�� L-1KMnO4����Һ�ζ����ﵽ�ζ��յ�ʱ�����ı���Һ20.00ml,�����������Ԫ�ص����������� ��



 �����Ϣ���£�
�����Ϣ���£� a.���Ȼ�����ˮ����ˮ�⣻
a.���Ȼ�����ˮ����ˮ�⣻ b.�������������ڸ����¾���������ֱ�ӷ�Ӧ������Ӧ���Ȼ��
b.�������������ڸ����¾���������ֱ�ӷ�Ӧ������Ӧ���Ȼ�� c.�й����ʵ������������±���
c.�й����ʵ������������±���

 ��ش��������⣺
��ش��������⣺ ��1��д��װ��A�з�����Ӧ�����ӷ���ʽ ��
��1��д��װ��A�з�����Ӧ�����ӷ���ʽ �� ��2��װ��A��g�ܵ������� ��װ��C�е��Լ��� ��װ��E�е�hƿ��Ҫ��ȴ������ ��
��2��װ��A��g�ܵ������� ��װ��C�е��Լ��� ��װ��E�е�hƿ��Ҫ��ȴ������ �� ��3��װ��E��hƿ�ռ����Ĵֲ����ͨ���������ƶ�����õ��ߴ������Ȼ��裬�����IJ������У�����Ԫ������ܻ����е�����Ԫ���� ����дԪ�ط��ţ���
��3��װ��E��hƿ�ռ����Ĵֲ����ͨ���������ƶ�����õ��ߴ������Ȼ��裬�����IJ������У�����Ԫ������ܻ����е�����Ԫ���� ����дԪ�ط��ţ��� ��4��Ϊ�˷�������������Ԫ�صĺ������Ƚ�������Ԥ����������Ԫ�ػ�ԭ��Fe2+������KMnO4����Һ�����������½���������ԭ�ζ�����Ӧ�����ӷ���ʽ�ǣ�
��4��Ϊ�˷�������������Ԫ�صĺ������Ƚ�������Ԥ����������Ԫ�ػ�ԭ��Fe2+������KMnO4����Һ�����������½���������ԭ�ζ�����Ӧ�����ӷ���ʽ�ǣ�
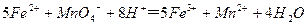
 �ٵζ�ǰ�Ƿ�Ҫ�μ�ָʾ���� ����ǡ�������˵������ ��
�ٵζ�ǰ�Ƿ�Ҫ�μ�ָʾ���� ����ǡ�������˵������ �� ��ijͬѧ��ȡ5.000g�����Ԥ������������ƿ�����Ƴ�100ml��Һ����ȡ25.00ml��������Һ����1.000��10-2mol�� L-1KMnO4����Һ�ζ����ﵽ�ζ��յ�ʱ�����ı���Һ20.00ml,�����������Ԫ�ص����������� ��
��ijͬѧ��ȡ5.000g�����Ԥ������������ƿ�����Ƴ�100ml��Һ����ȡ25.00ml��������Һ����1.000��10-2mol�� L-1KMnO4����Һ�ζ����ﵽ�ζ��յ�ʱ�����ı���Һ20.00ml,�����������Ԫ�ص����������� ����1��MnO2 + 4H�� + 2Cl��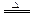 Mn2�� + Cl2�� + 2H2O
Mn2�� + Cl2�� + 2H2O
 ��2��ƽ��ѹǿ Ũ���� ʹSiCl4����
��2��ƽ��ѹǿ Ũ���� ʹSiCl4����
 ��3��Al��P��Cl
��3��Al��P��Cl
 ��4���ٷ� KMnO4��Һ��������ָʾ����
��4���ٷ� KMnO4��Һ��������ָʾ����
 ��4.480%
��4.480%
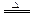 Mn2�� + Cl2�� + 2H2O
Mn2�� + Cl2�� + 2H2O ��2��ƽ��ѹǿ Ũ���� ʹSiCl4����
��2��ƽ��ѹǿ Ũ���� ʹSiCl4���� ��3��Al��P��Cl
��3��Al��P��Cl ��4���ٷ� KMnO4��Һ��������ָʾ����
��4���ٷ� KMnO4��Һ��������ָʾ���� ��4.480%
��4.480%��

��ϰ��ϵ�д�
 һ����ʦȨ����ҵ��ϵ�д�
һ����ʦȨ����ҵ��ϵ�д�
�����Ŀ
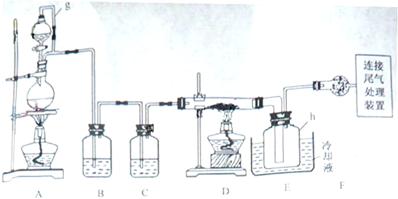
 ��ͬ��Ϊ����֤��һ��ʵ��ijͬѧ������ͼװ�ý���ʵ�飨����ʵ������ΪѹǿΪ101 kPa���¶�Ϊ0�棩��
��ͬ��Ϊ����֤��һ��ʵ��ijͬѧ������ͼװ�ý���ʵ�飨����ʵ������ΪѹǿΪ101 kPa���¶�Ϊ0�棩��
 g����Բ����ƿ�з�����Ӧ�Ļ�ѧ����ʽΪ�� ��
g����Բ����ƿ�з�����Ӧ�Ļ�ѧ����ʽΪ�� �� �ܱ����С�����������
�ܱ����С�����������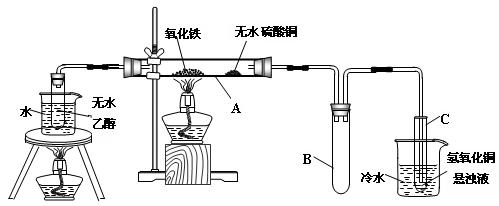
 2��ʵ���пɹ۲쵽ʯӢ��A�е�����Ϊ ��
2��ʵ���пɹ۲쵽ʯӢ��A�е�����Ϊ ��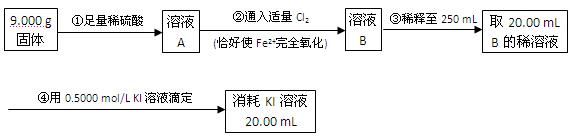
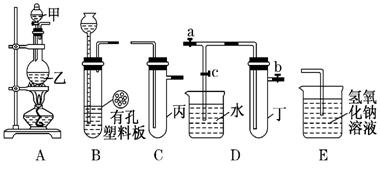
 ��װ�ÿ�������ȡCl2��������ص�����ʵ�飮
��װ�ÿ�������ȡCl2��������ص�����ʵ�飮 _____________________________.
_____________________________. OC�DCOOH���ɼ�дΪH2C2O4)�׳Ʋ��ᣬ������ˮ�����ڶ�Ԫ��ǿ��(Ϊ�������)��������ǿ��̼�ᣬ���۵�Ϊ101.5�棬��157��������ijУ�о���ѧϰС��Ϊ̽������IJ��ֻ�ѧ���ʣ�����������ʵ�飺
OC�DCOOH���ɼ�дΪH2C2O4)�׳Ʋ��ᣬ������ˮ�����ڶ�Ԫ��ǿ��(Ϊ�������)��������ǿ��̼�ᣬ���۵�Ϊ101.5�棬��157��������ijУ�о���ѧϰС��Ϊ̽������IJ��ֻ�ѧ���ʣ�����������ʵ�飺 HCO3��Һ���Թ��м��������Ҷ�����Һ���۲쵽����ɫ���ݲ������÷�Ӧ�����ӷ���ʽΪ_______
HCO3��Һ���Թ��м��������Ҷ�����Һ���۲쵽����ɫ���ݲ������÷�Ӧ�����ӷ���ʽΪ_______ __________________________________��
__________________________________��