��Ŀ����
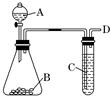
(13��) ������ͼװ�ý���ͭ��һ�������֪Ũ�ȵ�Ũ���ᷴӦ��ʵ���о���
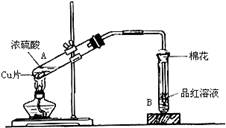
��1��д���Թ�B�е�ʵ������ ��
��2��д��A����Ӧ�Ļ�ѧ����ʽ������˫���ű�������ת�Ƶķ������Ŀ�� ��
��3����ַ�Ӧ��ѧ������ͭ�����ᶼ��ʣ�ࡣ�ڲ�����Ũ�����ǰ���£���Ҫʹʣ���ͭƬ�ܽ⣬���ټ��루Ҫ����дһ�ֲ�����������ʣ� ��
��4��B�Թܿڵ���Ӧմ�е��Լ��� ���������� �����ӷ���ʽΪ ��

��1��д���Թ�B�е�ʵ������ ��
��2��д��A����Ӧ�Ļ�ѧ����ʽ������˫���ű�������ת�Ƶķ������Ŀ�� ��
��3����ַ�Ӧ��ѧ������ͭ�����ᶼ��ʣ�ࡣ�ڲ�����Ũ�����ǰ���£���Ҫʹʣ���ͭƬ�ܽ⣬���ټ��루Ҫ����дһ�ֲ�����������ʣ� ��
��4��B�Թܿڵ���Ӧմ�е��Լ��� ���������� �����ӷ���ʽΪ ��
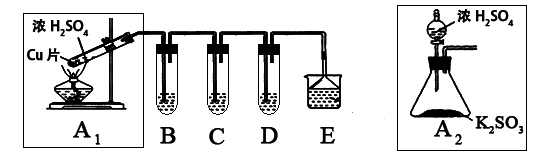
��1��Ʒ����Һ��ɫ
��2��
��3��NaNO3��Fe2O3��FeCl3�����������𰸾��ɣ�
��4��NaOH��Һ ����β������ֹ��Ⱦ 2OH����SO2��SO32����H2O
��2��

��3��NaNO3��Fe2O3��FeCl3�����������𰸾��ɣ�
��4��NaOH��Һ ����β������ֹ��Ⱦ 2OH����SO2��SO32����H2O
�����������1���Թ�B�е�������Ʒ����Һ��ɫ����2��A����Ӧ�Ļ�ѧ����ʽ��˫����Ϊ��
 ����3��ͭ��Ũ���ᷴӦʱ��������ϡ��һ���̶�ʱ���÷�Ӧֹͣ���������ͭ�����ܽ���Լ�������������NaNO3��Fe2O3��FeCl3�ȡ���4��SO2�Ǵ�����Ⱦ�����Ӧ����NaOH��Һ���գ���Ӧ�����ӷ���ʽΪ��2OH-+SO2=SO32-+H2O��
����3��ͭ��Ũ���ᷴӦʱ��������ϡ��һ���̶�ʱ���÷�Ӧֹͣ���������ͭ�����ܽ���Լ�������������NaNO3��Fe2O3��FeCl3�ȡ���4��SO2�Ǵ�����Ⱦ�����Ӧ����NaOH��Һ���գ���Ӧ�����ӷ���ʽΪ��2OH-+SO2=SO32-+H2O����������ʵ��Ƚϻ������Ѷ�С������Ϊ��ѧ�����л���ѵ����ϰ�⡣

��ϰ��ϵ�д�
 ������������ϵ�д�
������������ϵ�д�
�����Ŀ