��Ŀ����
(I)���ӹ�ҵ����30����FeCl3����Һ��ʴ���ھ�Ե���ϵ�ͭ��������ӡˢ��·�塣��FeCl3��Һ����ʴҺ��Cu��Ӧ����CuCl2��FeCl2��
(1)д���÷�Ӧ�Ļ�ѧ����ʽ ��
(2)������Һ��Fe3+���ڵ��Լ���
֤��Fe2+���ڵ������� ��
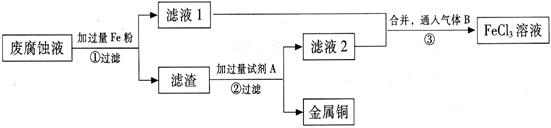
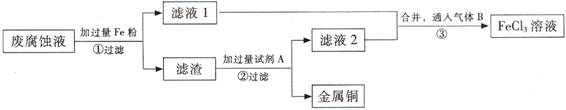
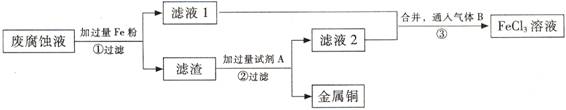
(��)ӡˢ��·�ķϸ�ʴҺ���д���CuCl2��FeCl2��FeCl3�������ŷŻ���ɻ�����Ⱦ����Դ���˷ѡ�ͨ���������̿ɴӸ÷�Һ�л���ͭ���������Ļ�����ȫ��ת��ΪFeCl3 ��Һ����Ϊ��ʴҺԭ��ѭ��ʹ�á�

(3) ������з�����Ӧ�����ӷ�
(4)��������Ҫ�ɷ��� (�ѧʽ)��
(5)Ϊ�˳�ȥ�����е����ʵõ�ͭ���������Լ�A�� (�ѧʽ)��
(6)�����ͨ�˵�����B (�ѧʽ)��
д���÷�Ӧ�Ļ�ѧ����ʽ ��
(I)
(1)2FeCl3+Cu==2FeCl2��CuCl2 (2��)
[��ѧʽ�������֣�û����ƽ��1��]
(2)KSCN��Һ(1��)�� ��Һ���Ѫ��ɫ(1��)
(II)
(3)2Fe3+++Fe��3Fe2+ (1��) Fe+Cu2+��Fe2++Cu (1��) ![]()
����ѧʽ�������֣�û����ƽ��1�֣�
(4)Cu��Fe (1��)
(5)HCI (1��)
(6)C12 (1��) 2FeCl2+C12��2FeCl3 (2��) 

 (3) ������з�����Ӧ�����ӷ���ʽ
(3) ������з�����Ӧ�����ӷ���ʽ 
 (3) ������з�����Ӧ�����ӷ���ʽ
(3) ������з�����Ӧ�����ӷ���ʽ