��Ŀ����
2��ʵ�����Կ�����O2�������20%��Ϊԭ�ϣ�����̼����ˮ�Ļ����£�����ͼAװ���Ʊ�������3O2$\frac{\underline{\;�ŵ�\;}}{\;}$2O3����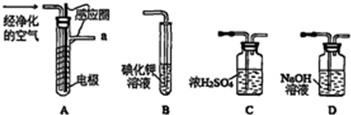
��1������ͨ��Aװ��֮ǰ��Ӧ�Ⱥ�ͨ������װ���е�D��C����װ����ţ���
��2��������⻯����Һ��ӦΪ��2KI+03+H20=2KOH+I2+02����a������ͨ��װ��B����Һ�е�����Ϊ��Һ�����Ϻ�ɫ
��3��Ϊ�ⶨ02ת��Ϊ03��ת���ʣ���װ��B�е���Һȫ��ת����һ�����У�����CC14������ȡ����Һ��������ȴ�����أ���I2����0.254g��
����ȡ�������ò������������Ʒ�Һ©��
����ʵ��ʱͨ�����1.12L����״������O2��ת����Ϊ15%
�۲ⶨʱ����A��Bװ�ü�����װ��D��ԭ���dz�ȥ�����еĵ���������
��4����ҵ�Ϸ���03��02���ɽ��������Һ�����ٷ��룬���з��뷽����������B������ţ���
A�����ˡ�B������C����Һ��D����ȡ
��5�����������ں�CNһ���Ե�Ʒ�ˮ�Ĵ�������i����CNһת��ΪOCN-����ii����OCNһ����ת��ΪCO32һ�����ֵ������壮����ii��ת��ʱ��O3��OCN-���ʵ�����֮��Ϊ3��2���ò���Ӧ�����ӷ���ʽΪ2OH-+3O3+2OCN-=2CO32-+N2+3O2+H2O��
���� ��1��O2����̼����ˮ�Ļ������Ʊ����������Կ����е�ˮ�Ͷ�����̼Ҫ�ȳ�ȥ��
��2��03���⻯�������ɵ⣬�ݴ��жϵ⻯����Һ����ɫ�仯��
��3������ȡʵ����Ҫ�÷�Һ©����
�ڸ��ݹ�ϵʽI2��03��$\frac{3}{2}$O2���ɼ����ת���˵����������ʵ���������ת����=$\frac{��ת�������������ʵ���}{��ʼ���������ʵ���}$��100%���㣻
���ڷŵ������£������е������п����뵪�����������������Щ�����������������⻯�أ�����Ҫ��ȥ��
��4������03��02�ķе㲻ͬ�����Կɽ��������Һ�����ٷ���ķ������з��룻
��5��OCNһ����ת��ΪCO32һ�����ֵ������壬�����ֵ���ӦΪ��������������O3��OCN-���ʵ�����֮��Ϊ3��2ʱ�����ݵ���غ��д�����ӷ���ʽ��
��� �⣺��1��O2����̼����ˮ�Ļ������Ʊ������������е�ˮ�Ͷ�����̼Ҫ�ȳ�ȥ�����Կ���ͨ��Aװ��֮ǰ��Ӧ�Ⱥ�ͨ������װ���е�D��C��
�ʴ�Ϊ��D��C��
��2��03���⻯�������ɵ⣬��ɫ�ĵ⻯����Һ������Ϻ�ɫ���ʴ�Ϊ����Һ�����Ϻ�ɫ��
��3������ȡʵ����Ҫ�÷�Һ©�����ʴ�Ϊ����Һ©����
�ڵ�I2����0.254g��0.001molʱ�����ݹ�ϵʽI2��03��$\frac{3}{2}$O2����֪�μӷ�Ӧ�����������ʵ���Ϊ0.0015mol��ʵ��ʱͨ�����1.12L��0.05mol���������������ʵ���Ϊ0.01mol������O2��ת����Ϊ$\frac{0.0015mol}{0.01mol}$��100%=15%��
�ʴ�Ϊ��15%��
���ڷŵ������£������е������п����뵪�����������������Щ�����������������⻯�أ����Բⶨʱ����A��Bװ�ü�����װ��D��
�ʴ�Ϊ����ȥ�����еĵ��������
��4������03��02�ķе㲻ͬ�����Կɽ��������Һ�����ٷ���ķ������з��룬��ѡB��
��5��OCN-����ת��ΪCO32-�����ֵ������壬�����ֵ���ӦΪ��������������O3��OCN-���ʵ�����֮��Ϊ3��2ʱ����Ӧ�����ӷ���ʽΪ2OH-+3O3+2OCN-=2CO32-+N2+3O2+H2O��
�ʴ�Ϊ��2OH-+3O3+2OCN-=2CO32-+N2+3O2+H2O��
���� ������Ҫ���������ʵ��Ʊ������Ӻ��ᴿ���Ѷ��еȣ�ע��������Ŀ��Ϣ�������⣬������ѧ����������������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ������ĵ���ʽ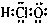 | B�� | ��ϩ�Ľṹ��ʽ��C2H4 | ||
| C�� | ������̼�ı���ģ�ͣ� | D�� | Cl-�Ľṹʾ��ͼ��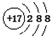 |
| ʵ�� | ҩƷ | ��ȡ���� | �������е�Һ�� |
| �� | Cu��ϡHNO3 | H2O | |
| �� | NaOH���塢Ũ��ˮ | NH3 | |
| �� | Na2CO3���塢ϡH2SO4 | CO2 | |
| �� | þ���Ͻ�NaOH��Һ�������� | H2 | H2O |
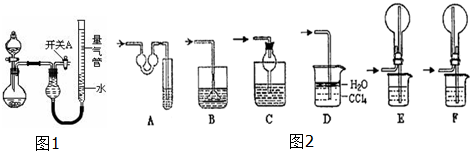
��1��������μ���װ�õ������ԣ��������м�ˮ��رտ���A�ͷ�Һ©������������ƿ���������Ҷ�Һ�������ұ��ֲ��䣬˵�������Ժã�
��2����ͬѧ��Ϊʵ��ٿ�ͨ���ռ�����NO������������̽��Cu��Ʒ�Ĵ��ȣ�����Ϊ�Ƿ���У������ԭ�����У���ΪNO����װ���п�����Ӧ������ˮ��ʹ��õ�NO�����������
��3��ʵ�����ʣ���NH3�����մ��������¸���β������װ���У��ʺ�������NH3�������ܷ�ֹ��������ACDF
��4��ʵ����У��������е�Һ�������D��
A��H2O������B��CCl4��������C������Na2CO3��Һ���� D������NaHCO3��Һ
��5����ʵ��Ӧ�������ܶ�ζ���������ʱӦע�⣺
�ٻָ������£���ʹ����������Һ����ƽ���������밼Һ����ʹ���ƽ��
��6��ʵ��ܻ���������ݣ���������������ѻ���ɱ�״����
| ��� | þ���Ͻ����� | �����ܵ�һ�ζ��� | �����ܵڶ��ζ��� |
| �� | 1.0g | 10.0mL | 346.3mL |
| �� | 1.0g | 10.0mL | 335.0mL |
| �� | 1.0g | 10.0mL | 345.7mL |
| A�� | �û��������Է���������62 | |
| B�� | �û�����Ļ�ѧʽΪC2H6O2 | |
| C�� | 1mol�û���������2molO2������������Ӧ | |
| D�� | 1mol���л����������2molNa��Ӧ |
| ������ | ����K+������Na+���� Cu2+����Al3+ |
| ������ | ����SO42-����HCO3-����NO3-����OH- |
�ٽ���������ˮ��DΪ��ɫ��Һ��������Ϊ��ɫ��Һ��
�ڽ�E��Һ���뵽C��Һ�г��ְ�ɫ�����������μӣ������ܽ⣻
�۽�����ɫ��Ӧ��ֻ��B��CΪ��ɫ������ɫ�ܲ���Ƭ����
���ڸ���Һ�м������ᱵ��Һ���ټӹ���ϡ���ᣬA�зų���ɫ���壬C��D�в�����ɫ������
�ݽ�B��D����Һ��ϣ�δ���������������ɣ�
��������ʵ����գ�
��1��д��B��D�Ļ�ѧʽ��B��KNO3��D��CuSO4��
��2������1mol A����Һ�뺬1mol E����Һ��Ӧ�����ɣ����õ�һ�ֻ�����û�����ΪNa2CO3 ��
��3��д��ʵ��ڷ�����Ӧ�����ӷ���ʽ��Al3++3OH-�TAl��OH��3��Al��OH��3+OH-�T[Al��OH��4]-��
��4��C��������ˮ���������ӷ���ʽ��ʾ�侻ˮԭ����Al3++3H2O?Al��OH��3�����壩+3H+��
| A�� | ֻ�н������ʲ�����ɫ��Ӧ | B�� | ֻ�н��������������ɫ��Ӧ | ||
| C�� | ijЩ�������仯���ﶼ����ɫ��Ӧ | D�� | ֻ�н������Ӳ�����ɫ��Ӧ |
| A�� | ﮣ�Li����ˮ��Ӧ������ˮ��Ӧ���� | |
| B�� | ����At��Ϊ��ɫ���壬AgAt������ˮҲ������ϡ���� | |
| C�� | �������У�﨣�Rb����ȼ�ղ�����Ƶ�ȼ�ղ�������� | |
| D�� | HBrO4�����Ա�HClO4�� |
| A�� | NaOH��Һ��CO2�ķ�Ӧ | B�� | NaOH��Һ�ʹ���ķ�Ӧ | ||
| C�� | NaOH��Һ�����ᷴӦ | D�� | ��ˮ��ϡH2SO4�ķ�Ӧ |
