ΧβΡΩΡΎ»ί
œ¬ΆΦ « Β―ι “÷Τ±Η¬»Τχ≤ΔΫχ––“ΜœΒΝ–œύΙΊ Β―ιΒΡΉΑ÷ΟΘ®Φ–≥÷ΦΑΦ”»»“«Τς“―¬‘Θ©ΓΘ
Θ®1Θ©÷Τ±Η¬»Τχ―Γ”ΟΒΡ“©ΤΖΈΣΙΧΧεΕΰ―θΜ·ΟΧΚΆ≈®―ΈΥαΘ§‘ρœύΙΊΒΡάκΉ”Ζ¥”ΠΖΫ≥Χ ΫΈΣ ΘΜ
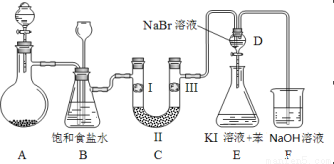
ΉΑ÷ΟB÷–±ΞΚΆ ≥―ΈΥ°ΒΡΉς”Ο «_________ΘΜΆ§ ±ΉΑ÷ΟB“ύ «Α≤»ΪΤΩΘ§Φύ≤β Β―ιΫχ–– ±C÷– «ΖώΖΔ…ζΕ¬»ϊΘ§«κ–¥≥ωΖΔ…ζΕ¬»ϊ ±B÷–ΒΡœ÷œσ ΘΜ
Θ®2Θ©ΉΑ÷ΟCΒΡ Β―ιΡΩΒΡ «―ι÷Λ¬»Τχ «ΖώΨΏ”–Τ·ΑΉ–‘Θ§ΈΣ¥ΥC÷–IΓΔIIΓΔIII“ά¥ΈΖ≈»κ ΘΜ
| a | b | c | d |
I | Η…‘οΒΡ”–…Ϊ≤ΦΧθ | Η…‘οΒΡ”–…Ϊ≤ΦΧθ | Σ»σΒΡ”–…Ϊ≤ΦΧθ | Σ»σΒΡ”–…Ϊ≤ΦΧθ |
II | Φν ·Μ“ | ΙηΫΚ | ≈®ΝρΥα | ΈόΥ°¬»Μ·ΗΤ |
III | Σ»σΒΡ”–…Ϊ≤ΦΧθ | Σ»σΒΡ”–…Ϊ≤ΦΧθ | Η…‘οΒΡ”–…Ϊ≤ΦΧθ | Η…‘οΒΡ”–…Ϊ≤ΦΧθ |
Θ®3Θ©…ηΦΤΉΑ÷ΟDΓΔEΒΡΡΩΒΡ «±»Ϋœ¬»ΓΔδεΓΔΒβΒΞ÷ ΒΡ―θΜ·–‘«Ω»θΓΘΒ±œρD÷–ΜΚΜΚΆ®»κ…ΌΝΩ¬»Τχ ±Θ§Ω…“‘Ω¥ΒΫΈό…Ϊ»ή“Κ÷πΫΞ±δΈΣ__________…ΪΘ§ΥΒΟς ΘΜ
¥ρΩΣΜν»ϊΘ§ΫΪΉΑ÷ΟD÷–…ΌΝΩ»ή“ΚΦ”»κΉΑ÷ΟE÷–Θ§’ώΒ¥ΓΘΙέ≤λΒΫΒΡœ÷œσ « ΘΜ
Θ®4Θ©ΉΑ÷ΟF÷–”ΟΉψΝΩΒΡNaOH»ή“ΚΈϋ ’”ύ¬»Θ§ ‘–¥≥ωœύ”ΠΒΡάκΉ”ΖΫ≥Χ Ϋ ΘΜ
Θ®1Θ©MnO2+4H++2Cl? Mn2++Cl2Γϋ+ 2H2OΘ®2Ζ÷Θ©ΘΜ
Mn2++Cl2Γϋ+ 2H2OΘ®2Ζ÷Θ©ΘΜ
≥ΐ»ΞCl2÷–ΒΡHClΘ®1Ζ÷Θ©ΘΜ B÷–≥ΛΨ±¬©ΕΖ÷–“ΚΟφ…œ…ΐΘ§–Έ≥…Υ°÷υΘ®2Ζ÷Θ©ΘΜ
Θ®2Θ©dΘ®2Ζ÷Θ©ΘΜ
Θ®3Θ©ΜΤΘ®1Ζ÷Θ©ΘΜ¬»ΤχΒΡ―θΜ·–‘«Ω”Ύδε Θ®2Ζ÷Θ© E÷–»ή“ΚΖ÷ΈΣΝΫ≤ψΘ§…œ≤ψΘ®±Ϋ≤ψΘ©ΈΣΉœΚλ…ΪΘ®2Ζ÷Θ©ΘΜ
Θ®4Θ©2OHΓΣ+Cl2 ΘΫClΓΣ+ClOΓΣ+H2OΘ®2Ζ÷Θ©
ΓΨΫβΈωΓΩ
‘ΧβΖ÷ΈωΘΚΘ®1Θ©”…Μ·―ßΖΫ≥Χ ΫΗΡ–¥Ω…ΒΟάκΉ”ΖΫ≥Χ ΫΘΜ±ΞΚΆ ≥―ΈΥ°Ω…Έϋ ’HClΘ§ΉΑ÷ΟB÷–±ΞΚΆ ≥―ΈΥ°ΒΡΉς”Ο «≥ΐ»ΞCl2÷–ΒΡHClΘΜ»τΉΑ÷ΟCΕ¬»ϊΘ§B÷–―Ι«Ω‘ω¥σΘ§≥ΛΨ±¬©ΕΖ÷–“ΚΟφΜα…œ…ΐΘ§–Έ≥…Υ°÷υΓΘ
Θ®2Θ©ΉΑ÷ΟCΒΡ Β―ιΡΩΒΡ «―ι÷Λ¬»Τχ «ΖώΨΏ”–Τ·ΑΉ–‘Θ§“Σ―ι÷ΛΗ…‘כּΤχΈόΤ·ΑΉ–‘Ω…”Ο Σ»σΒΡ”–…Ϊ≤ΦΧθΘ§¬»ΤχΚΆΥ°Ζ¥”Π…ζ≥…¥Έ¬»ΥαΨΏ”–Τ·ΑΉ–‘Θ§―Γœν÷–abcΒΡΔρ÷–ΕΦ «Η…‘οΦΝΘ§‘ΌΆ®»κ Σ»σΒΡ”–…Ϊ≤ΦΧθ≤ΜΡή―ι÷Λ¬»ΤχΒΡΤ·ΑΉ–‘Θ§Υυ“‘C÷–IΓΔIIΓΔIII“ά¥ΈΖ≈»κ Σ»σΒΡ”–…Ϊ≤ΦΧθΓΔΈόΥ°¬»Μ·ΗΤΓΔΗ…‘οΒΡ”–…Ϊ≤ΦΧθΘ§Υυ“‘―ΓdΓΘ
Θ®3Θ©Β±œρD÷–ΜΚΜΚΆ®»κ…ΌΝΩ¬»Τχ ±Θ§Ω…“‘Ω¥ΒΫΈό…Ϊ»ή“Κ÷πΫΞ±δΈΣΜΤ…ΪΘ§ΥΒΟς¬»ΤχΚΆδεΜ·ΡΤΖ¥”Π…ζ≥…δεΒΞ÷ Θ§ΖΔ…ζΖ¥”ΠCl2+2NaBr=Br2+2NaClΘ§Ζ¥”Π÷–¬»Τχ «―θΜ·ΦΝΘ§δε Ι―θΜ·≤ζΈοΘ§Ι ―θΜ·–‘Br2ΘΦCl2ΓΘ¥ρΩΣΜν»ϊΘ§ΫΪΉΑ÷ΟD÷–Κ§δεΒΞ÷ ΒΡ…ΌΝΩ»ή“ΚΦ”»κλ ΒβΜ·ΦΊΚΆ±ΫΒΡΉΑ÷ΟE÷–Θ§δεΒΞ÷ ΚΆΒβΜ·ΦΊΖ¥”Π…ζ≥…ΒβΒΞ÷ Θ§ΒβΒΞ÷ »ή”Ύ±Ϋ≥ ΉœΚλ…ΪΘ§’ώΒ¥Θ°Ιέ≤λΒΫΒΡœ÷œσ «ΘΚE÷–»ή“ΚΖ÷ΈΣΝΫ≤ψΘ§…œ≤ψΘ®±Ϋ≤ψΘ©ΈΣΉœΚλ…ΪΓΘ
Θ®4Θ©Cl2”κNaOHΖ¥”Π…ζ≥…NaClΓΔNaClOΓΔH2OΘ§ΗΡ–¥Ω…ΒΟάκΉ”ΖΫ≥Χ ΫΓΘ
ΩΦΒψΘΚ±ΨΧβΩΦ≤鬻Τχ Β―ι “÷Τ»ΓΓΔ¬»ΤχΜ·―ß–‘÷ ΓΔ Β―ι…ηΦΤΓΔ Β―ιΉΑ÷ΟΒΡάμΫβΤάΦέΓΔΖΫ≥Χ ΫΒΡ ι–¥ΓΘ

 “ΜΨμΗψΕ®œΒΝ–¥πΑΗ
“ΜΨμΗψΕ®œΒΝ–¥πΑΗ Οϊ–ΘΉς“Β±ΨœΒΝ–¥πΑΗ
Οϊ–ΘΉς“Β±ΨœΒΝ–¥πΑΗ «α«…ΕαΙΎ÷ή≤⑬ΩΦ÷±Ά®Οϊ–ΘœΒΝ–¥πΑΗ
«α«…ΕαΙΎ÷ή≤⑬ΩΦ÷±Ά®Οϊ–ΘœΒΝ–¥πΑΗ