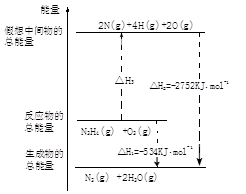��Ŀ����
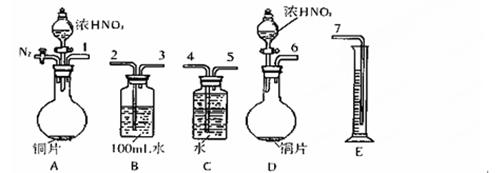
��ͼ��ʾ����ʵ���ҽ��а��������Ʊ�������ʵ������װ�ã����̶ֹ�װ��δ������
��l��д��װ��A���������Ļ�ѧ��Ӧ
��2��װ��B��ʢ���Լ���
��3����ȼC���ƾ��ƣ��رյ��ɼ�2�����ɼ�1���ӷ�Һ©���ų�Ũ��ˮ����û��ƿ�й����رշ�Һ©�����Ժ�Ƭ�̣�װ��C�к�ɫ������죬װ��E����Һ����ִ������ݣ�ͬʱ��
�� ����д��Ӧ������E���ݳ�Һ����������ֱ�������������д����C�з�����Ӧ�Ļ�ѧ����ʽ
��4����֪Cu2O��һ�ֺ�ɫ��������, �ڸ��������¿���CuO�ֽ�õ���4CuO��2Cu2O+O2�������ɵ�Cu2O Ҳ�ܱ�NH3��ԭ����C�й���ȫ�����ɫ�رյ��ɼ�1�������ƿ��ƾ��ƣ�����ȴ����C�й�������������Ӧǰ��������Ϊ16g����Ӧ����ع�����������2��4g��ͨ������ȷ���ù������ijɷ��� ���û�ѧʽ��ʾ��
��5���ڹرյ��ɼ�1���ɼ�2�������������F�У��ܿ췢��װ��F�в������̣�ͬʱ����G����ҺѸ�ٵ�������F�С�д���������̵Ļ�ѧ����ʽ
��1��NH3��H2O��CaO�� Ca(OH)2 + NH3 �� ��2����ʯ�һ���ʯ��
��3����ɫ������2NH3��3CuO 3Cu+N2����3H2O ��4��Cu2O��Cu
3Cu+N2����3H2O ��4��Cu2O��Cu
��5��3Cl2��8NH3��N2��6NH4Cl
���������������l��װ��AΪ�����ķ���װ�ã�����Ũ��ˮ����ʯ�ҷ�Ӧ�����������ƺͰ����������Ļ�ѧ��ӦΪNH3��H2O��CaO�� Ca(OH)2 + NH3 ������2��װ��BΪ�����ĸ���װ�ã�ʢ���Լ��Ǽ�ʯ�һ���ʯ�ң� ��3��װ��E������Ϊ��ȥ�����İ����������ķ�ӦΪSO2+2NH3��H2O����NH4��2SO3+H2O��
��NH4��2SO3+BaCl2 Ba SO3+ NH4Cl��װ��E�е�����Ϊ����Һ����ִ������ݣ�ͬʱ������ɫ��������������֪��װ��C�з����ķ�ӦΪ����������ͭ�ڼ��������·�Ӧ����ͭ��������ˮ����ѧ����ʽΪ2NH3��3CuO
Ba SO3+ NH4Cl��װ��E�е�����Ϊ����Һ����ִ������ݣ�ͬʱ������ɫ��������������֪��װ��C�з����ķ�ӦΪ����������ͭ�ڼ��������·�Ӧ����ͭ��������ˮ����ѧ����ʽΪ2NH3��3CuO 3Cu+N2����3H2O����4��16gCuO�к���ͭԪ������Ϊ16g��64/80=12.8g��������Ԫ������Ϊ16g-12.8g=3.2g����Ӧ����ع�����������2.4g��ʣ���������Ϊ16g-2.4g=13.6g������12.8g����ʣ����庬��Cu��OԪ�أ�n��Cu��=0.2mol��������Ԫ�ص�����Ϊ13.6g-12.8g=0.8g��n��O��=0.05mol��n��Cu����n��O��=0.2mol��0.05mol=4��1��2��1��ʣ�����ΪCu2O��Cu����5���ڹرյ��ɼ�1���ɼ�2�������������F�У�װ��F�в������̣������ķ�ӦΪ3Cl2��8NH3��N2��6NH4Cl��
3Cu+N2����3H2O����4��16gCuO�к���ͭԪ������Ϊ16g��64/80=12.8g��������Ԫ������Ϊ16g-12.8g=3.2g����Ӧ����ع�����������2.4g��ʣ���������Ϊ16g-2.4g=13.6g������12.8g����ʣ����庬��Cu��OԪ�أ�n��Cu��=0.2mol��������Ԫ�ص�����Ϊ13.6g-12.8g=0.8g��n��O��=0.05mol��n��Cu����n��O��=0.2mol��0.05mol=4��1��2��1��ʣ�����ΪCu2O��Cu����5���ڹرյ��ɼ�1���ɼ�2�������������F�У�װ��F�в������̣������ķ�ӦΪ3Cl2��8NH3��N2��6NH4Cl��
���㣺���鰱�����Ʊ�������ʵ�顣

 Сѧ�����ҵ���ϴ�ѧ������ϵ�д�
Сѧ�����ҵ���ϴ�ѧ������ϵ�д� ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д� �����������ҵ�������������ϵ�д�
�����������ҵ�������������ϵ�д� ѧ���������ν��Ͼ���ѧ������ϵ�д�
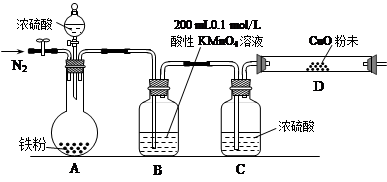
ѧ���������ν��Ͼ���ѧ������ϵ�д� (12��)ij�о�С����̽��SO2�Ļ�ѧ���ʣ����������ʵ�鷽����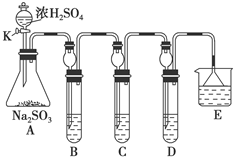
��1����B�м���SO2�������ԣ���B����ʢ�Լ���Ϊ________��
��2����C��װFeCl3��Һ������SO2�Ļ�ԭ�ԣ���C�з�Ӧ�����ӷ���ʽΪ__________________________��
��3����D��װ����Ư��Ũ��Һ��ͨ��SO2һ��ʱ���D�г����˴�����ɫ������ͬѧ�Ƕ�ɫ�����ɷֽ�����̽������ش��������⣺
��ѡ���������Լ�������װ�á��Թܡ��ιܡ������ܵĵ�����������ˮ��0��5 mol��L��1���ᡢ0��5 mol��L��1H2SO4��Һ��0��5 mol��L��1BaCl2��Һ��Ʒ����Һ�����Ƴ���ʯ��ˮ��
(��)����һ���ð�ɫ����ΪCaSO3��
��������ð�ɫ����Ϊ________��
���������ð�ɫ����Ϊ�����������ʵĻ���
(��)���ڼ���һ����д�±���
| ʵ����� | Ԥ������ͽ��� |
| ��D�г������ˣ�ϴ�Ӹɾ����� | |
| ����һ�ɾ��Թ�ȡ����������Ʒ������ ______ | __________ |
__________________________________________________��
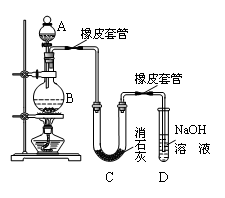
ij��ȤС����Ƴ���ͼ��ʾװ�����Ľ��̲��С�ͭ�����ᷴӦ��ʵ�飬��̽����ѧʵ�����ɫ����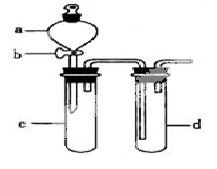
��1��ʵ��ǰ���رջ���b���Թ�d�м�ˮ����û�����ܿڣ������Թ�c��d�Ľ���������c����Ŀ����__________��
��2����d�м�����NaOH��Һ��c�з�һС��ͭƬ���ɷ�Һ©��a��c�м���2mLŨ���ᣬc�з�Ӧ�Ļ�ѧ����ʽ��______________________��
����a��c�м�2mL����ˮ��c�е�ʵ��������_____________��
��3���±�����ȡ����ͭ�����ַ�������������ɫ��ѧ�������ѷ�����_____��������_______��
| ���� | ��Ӧ�� |
| �� | Cu��Ũ���� |
| �� | Cu��ϡ���� |
| �� | Cu��O2��ϡ���� |