��Ŀ����
����Ŀ�������йص������Һ��˵����ȷ����( )
A.pH=1��һԪ���pH=13��һԪ��������Ϻ����Һ�У�һ���У�c(OH-)=c(H+)
B.���ô�����Һ��ϴ������ԭ���ǣ�CO32-+2H2O![]() H2CO3+2OH-
H2CO3+2OH-
C.�������ɲ�����AlCl3��Һ�õ�Al(OH)3����
D.�����£�Ksp(CaCO3)=2.8��10-9��Ksp(CaSO4)=9.1��10-6����CaCO3�ı�����Һ�м���Na2SO4��Ҳ���ܲ���CaSO4����
���𰸡�D
��������
A. �������ǿ�ᣬ����ǡ�÷�Ӧ�������Σ���Һ������c(OH)=c(H+)������������ᣬ���ߵ�������ʱ���������c(OH)<c(H+)����A����
B. ������ǿ�������Σ�ˮ����Һ�ʼ��ԣ��Ե�һ��ˮ��Ϊ��������֬�ڼ�������������ˮ�⣺CO32-��H2O![]() HCO3-��OH������B����
HCO3-��OH������B����
C. ��������AlCl3��Һʱ������ˮ�ⷴӦ����Al(OH)3������ʱ�ֽ�õ�Al2O3���壬��C����
D. �����£�Ksp(CaCO3)=2.8��10-9��Ksp(CaSO4)= 9.1��10-6����CaCO3�ı�����Һ�м���Na2SO4�����������Ũ�ȣ���Qc>Ksp(CaSO4)�ǣ�Ҳ���ܲ���CaSO4��������D��ȷ��
��ѡD��

����Ŀ��X��Y��Z��M��Q��R��6�ֶ�����Ԫ�أ���ԭ�Ӱ뾶����Ҫ���ϼ����£�
Ԫ�ش��� | X | Y | Z | M | Q | R |
ԭ�Ӱ뾶/nm | 0.160 | 0.143 | 0.102 | 0.089 | 0.074 | 0.037 |
��Ҫ���ϼ� | +2 | +3 | +6��-2 | +2 | -2 | +1 |
��1��Z��Ԫ�����ڱ��е�λ����______________________________��
��2��X��Y��Q�����γɼ����ӣ��������Ӱ뾶������__________�������ӷ��ţ���
��3�����ڱ�����Щ���ڶԽ��ߣ����ϡ����£�λ�õ�Ԫ�أ����ǵĵ��ʼ��仯������������ƣ���M����������ǿ����Һ��Ӧ�����ӷ���ʽ______________________________��
��4��Q��R��ԭ�Ӹ�����1:1��ɵĻ��������һ�֡���ɫ����������
�ټ����������ۼ�������____________________��
�ڿ�������������Ʊ���װ����ͼ��ʾ���ڼ�����Һ�У����ÿ����е�������ԭ�õ���ϡ�����Һ��ͼ��a��__________���������ĵ缫��Ӧʽ��____________________��

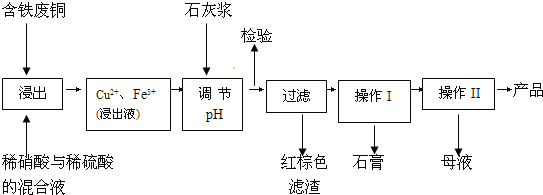
����Ŀ������ͭ��һ��Ӧ�ü���㷺�Ļ���ԭ�ϣ�������ij�����ú�����ͭΪԭ������������CuSO45H2O��������ʯ�ࣨCaSO42H2O������������ʾ��ͼ��
������ʯ���ڲ�ͬ�¶��µ��ܽ�ȣ�g/100gˮ��������
�¶ȣ��棩 | 20 | 40 | 60 | 80 | 100 |
ʯ�� | 0.32 | 0.26 | 0.15 | 0.11 | 0.07 |
���� | 32 | 44.6 | 61.8 | 83.8 | 114 |
��ش��������⣺
��1������ɫ��������Ҫ�ɷ���___��
��2��д��������������������ͭ�����ӷ���ʽ___��
��3������I��������Ũ����__�Ȳ������¶�Ӧ�ÿ�����__�����ң�
��4������Һ�з��������ͭ����IJ�����ӦΪ__��___��ϴ�ӡ������������ˮ�Ҵ���ϴ��Һ����������ˮ��ԭ����__������ʱ�������ɣ����ü��Ⱥ�ɵ�ԭ����___��
��5��ȡ��������Ϊ��ȷ��Fe3+�Ƿ��������ͬѧ������������ַ�������ʵ���ҷֱ����ȡ��Ʒ�����з������в�����
����һ��ȡ�����Թ����μ�KSCN��Һ�� ��������ֽ��������KSCN��Һ��
������Ϊ�������ַ�����Ƹ���������__��
��ָ������Ϊ�������������ڵ�������__��
����Ŀ��Ϊ�˲ⶨʵ���ҳ��ڴ�ŵ�Na2SO3����Ĵ��ȣ�ȷ��ȡM g������Ʒ�����250 mL��Һ���������������ʵ�鷽����
����I��ȡ50.00 mL������Һ�����������������ữ��BaCl2��Һ������I��ϴ�ӡ���������������õ�����������Ϊm1 g
������ȡ50.00 mL������Һ����a mol/L ������KMnO4��Һ���еζ���
ʵ��������¼���������±���
�ζ����� ʵ������ | 1 | 2 | 3 | 4 |
������Һ���/mL | 50.00 | 50.00 | 50.00 | 50.00 |
�ζ��ܳ�����/mL | 0.00 | 0.20 | 0.10 | 0.15 |
�ζ���ĩ����/mL | 20.95 | 21.20 | 20.15 | 21.20 |
(1)����250 mL Na2SO3��Һʱ�������õ���ʵ�������У��ձ����������ιܡ�ҩ��_______��________��
(2)����IΪ______________��������Ϊ______________��
(3)�ڷ������еζ��յ���жϷ�����_______________________________��
(4)�ڷ������з��������ӷ�Ӧ����ʽΪ____________________________��
(5)���ݷ��������ṩ�����ݣ�����Na2SO3�Ĵ���Ϊ___________����д�ɷ�����ʽ��
(6)��������������ԭ�ζ������У����´���ҺNa2SO3Ũ�ȱ�С����_____������ţ���
a���ü�ʽ�ζ�����ȡ50mL����Һ����ʱ����ʼ���ӣ��ζ�����ʱ����
b���ü�ʽ�ζ�����ȡ50mL����Һ����ʱ��һ��ʼ�����ݣ��ζ�������û����
c����ʽ�ζ���������ˮ��ϴ��û��������KMnO4��Һ�����ϴ
d����ƿ������ˮ��ϴ��ֱ��װ50.00mL�Ĵ���Һ
e���ζ�����ʱ����ʼʱƽ�ӣ��ζ�����ʱ����