��Ŀ����
����Ŀ���Ժ�ˮΪԭ�Ͽ�����ȡ����þ��ͼ��ij�����Ӻ�ˮ����ȡþ����Ҫ���裮��������������� 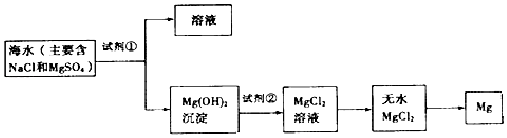
��1���ں�ˮ��þ�Ĺ��������ʵ�ֶ�þ���ӵĸ��� ������ѧ������Լ��Ĺ۵㣮 ѧ���Ĺ۵㣺ֱ������ˮ�м����������
ѧ���ҵĹ۵㣺����ɹ�κ�Ŀ�±ˮ���ټ����������
ѧ�����Ĺ۵㣺���¼���������ˮ���ټ����������
ͨ�������Ƚ�����Ϊѧ���Ĺ۵���ȷ����ѧ����ţ���
��2��Ϊ��ʹþ���ӳ���������������õ��صı��ǣ���Ҫ�ɷ�Ϊ̼��ƣ���Դ��ó�������������Լ��������ѧʽ����д���ɱ�����ȡ�Լ��ٵ���ػ�ѧ����ʽ����
��3�������Լ��ٺ��ܹ�����õ�Mg��OH��2�����ķ�������������ĸ��
A.����
B.��Һ
C.��ȡ
D.����
��4��������Լ��������ѧʽ������ҵ�����г����������ᾧ����Ȼ�þˮ���ᄃ��������HCl�����м��Ȼ����ˮMgCl2 �� ��Ŀ���� ��
��5��д������ˮMgCl2��ȡ����þ�Ļ�ѧ����ʽ ��
���𰸡�
��1����
��2��Ca��OH��2�� CaCO3![]() CaO+CO2��,CaO+H2O�TCa��OH��2
CaO+CO2��,CaO+H2O�TCa��OH��2
��3��D
��4��HCl������ˮ��,�õ���������ˮ�Ȼ�þ
��5��MgCl2���ۻ��� ![]() ?Mg+Cl2��
?Mg+Cl2��
���������⣺��1��ѧ�����������۵���ȣ���ˮ��þ����Ũ��С��ʹ�õij����������ϴ��Ҳ������ռ�������þ�����ѧ���Ĺ۵㲻��ȷ��ԭ���ǣ���ˮ��þ����Ũ��С��������������������þ���ӵij�����
ѧ������ѧ������ȣ�����ɹ�κ�Ŀ�±����Լ��Դ���ɱ��ͣ���ѧ�������þ����Ũ�ȸߣ�������þԪ�صĸ��������ѧ���ҵĹ۵���ȷ����Ϊ��þ���Ӹ���Ũ�ȸߣ��ɱ��ͣ�
ѧ�����������۵���ȣ�����������ˮ�����ĵ���Դ�࣬�ɱ�̫�ߣ����ѧ�����Ĺ۵㲻��ȷ��ԭ���ǣ���Դ���Ĵ�ˮ���ۺ����õͣ��ɱ��ߣ�
���Դ��ǣ��ң���2����̼��Ƹ��·ֽ�õ������ƣ���������ˮ��Ӧ�����������ƣ��������������Ȼ�þ��Ӧ����������þ��������Լ�����Ca��OH��2���ɱ�����ȡ�Լ��ٵ���ػ�ѧ����ʽ��CaCO3 ![]() CaO+CO2���� CaO+H2O�TCa��OH��2��
CaO+CO2���� CaO+H2O�TCa��OH��2��
���Դ��ǣ�Ca��OH��2�� CaCO3 ![]() CaO+CO2���� CaO+H2O�TCa��OH��2����3�������Լ��ٺ��ܹ�����õ�Mg��OH��2�����ķ����ǹ��˷��������ѡD��
CaO+CO2���� CaO+H2O�TCa��OH��2����3�������Լ��ٺ��ܹ�����õ�Mg��OH��2�����ķ����ǹ��˷��������ѡD��
���Դ��ǣ�D����4��Ҫʹ������þת��Ϊ�Ȼ�þ������Ҫ�������ᷴӦ�����Լ��������ᣬ��ҵ�����г����������ᾧ����Ȼ�þˮ���ᄃ��������HCl�����м��Ȼ����ˮMgCl2����Ŀ��������ˮ�⣬�õ���������ˮ�Ȼ�þ��
���Դ��ǣ�HCl������ˮ�⣬�õ���������ˮ�Ȼ�þ����5����ҵ��ȡþʱ���������״̬���Ȼ�þ��MgCl2 ![]() Mg+Cl2����
Mg+Cl2����
���Դ��ǣ���MgCl2 ![]() Mg+Cl2����
Mg+Cl2����