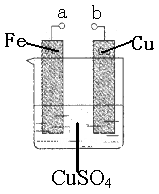
��Ŀ����
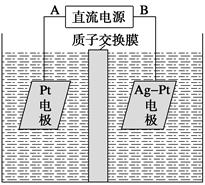
����ͼ��ʾװ�����ӣ�X��Y��Ϊ���Ե缫����ش��������⣺
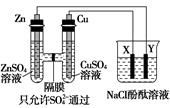
��1��ZnΪ________����
��2�����Ӻ�װ�ú��ձ��е���Һ������Ӧ�����ӷ���ʽ��___________��
��3��ͼ��ͨ����Ĥ��SO42-��________������ҡ�������Ǩ�ƣ�Y�����丽�����ֵ�������________��
��4�������£���Zn����������32.5 gʱ��X����������8.4 L����״����������ʱ�ձ�����Һ�����Ϊ500 mL�����ʱ�ձ�����Һ��pH��________�����������ɵ���������ˮ����

��1��ZnΪ________����
��2�����Ӻ�װ�ú��ձ��е���Һ������Ӧ�����ӷ���ʽ��___________��
��3��ͼ��ͨ����Ĥ��SO42-��________������ҡ�������Ǩ�ƣ�Y�����丽�����ֵ�������________��
��4�������£���Zn����������32.5 gʱ��X����������8.4 L����״����������ʱ�ձ�����Һ�����Ϊ500 mL�����ʱ�ձ�����Һ��pH��________�����������ɵ���������ˮ����
��1����
��2��2H2O��2Cl�� H2����Cl2����2OH��
H2����Cl2����2OH��
��3���������ݲ�������Һ�ʺ�ɫ
��4��14
��2��2H2O��2Cl��
 H2����Cl2����2OH��
H2����Cl2����2OH����3���������ݲ�������Һ�ʺ�ɫ
��4��14
��������֪�����Ϊԭ��أ��Ҳ�Ϊ���ء���1��ZnΪ��������2���ձ����ǵ���Ȼ�����Һ�����ӷ���ʽΪ2H2O��2Cl�� H2����Cl2����2OH������3��ZnΪ������ʧ��������Zn2��������Һ��SO42-ͨ����Ĥ�����ƶ����ձ���Y���������ݳ���ͬʱ��OH�����ɣ���̪��Һ�ʺ�ɫ����4��п��ԭ��صĸ�����Y�ǵ��ص�������ͭ��ԭ��ص�������X�ǵ��ص�������X�缫�������ݳ���ֱ��NaCl�����ȫ�����������ǵ��NaOH���ݳ�O2��Zn����0.5 mol��ת�Ƶ���Ϊ1 mol�����ȫ��������������0.5 mol�������ɣ����ȫ����������������0.25 mol�������ɡ����ݵ��ӵ�ʧ�غ����������ɵã�Cl2Ϊ0.25 mol��O2Ϊ0.125 mol�������֪��c��OH������1 mol/L��pH��14��
H2����Cl2����2OH������3��ZnΪ������ʧ��������Zn2��������Һ��SO42-ͨ����Ĥ�����ƶ����ձ���Y���������ݳ���ͬʱ��OH�����ɣ���̪��Һ�ʺ�ɫ����4��п��ԭ��صĸ�����Y�ǵ��ص�������ͭ��ԭ��ص�������X�ǵ��ص�������X�缫�������ݳ���ֱ��NaCl�����ȫ�����������ǵ��NaOH���ݳ�O2��Zn����0.5 mol��ת�Ƶ���Ϊ1 mol�����ȫ��������������0.5 mol�������ɣ����ȫ����������������0.25 mol�������ɡ����ݵ��ӵ�ʧ�غ����������ɵã�Cl2Ϊ0.25 mol��O2Ϊ0.125 mol�������֪��c��OH������1 mol/L��pH��14��
 H2����Cl2����2OH������3��ZnΪ������ʧ��������Zn2��������Һ��SO42-ͨ����Ĥ�����ƶ����ձ���Y���������ݳ���ͬʱ��OH�����ɣ���̪��Һ�ʺ�ɫ����4��п��ԭ��صĸ�����Y�ǵ��ص�������ͭ��ԭ��ص�������X�ǵ��ص�������X�缫�������ݳ���ֱ��NaCl�����ȫ�����������ǵ��NaOH���ݳ�O2��Zn����0.5 mol��ת�Ƶ���Ϊ1 mol�����ȫ��������������0.5 mol�������ɣ����ȫ����������������0.25 mol�������ɡ����ݵ��ӵ�ʧ�غ����������ɵã�Cl2Ϊ0.25 mol��O2Ϊ0.125 mol�������֪��c��OH������1 mol/L��pH��14��
H2����Cl2����2OH������3��ZnΪ������ʧ��������Zn2��������Һ��SO42-ͨ����Ĥ�����ƶ����ձ���Y���������ݳ���ͬʱ��OH�����ɣ���̪��Һ�ʺ�ɫ����4��п��ԭ��صĸ�����Y�ǵ��ص�������ͭ��ԭ��ص�������X�ǵ��ص�������X�缫�������ݳ���ֱ��NaCl�����ȫ�����������ǵ��NaOH���ݳ�O2��Zn����0.5 mol��ת�Ƶ���Ϊ1 mol�����ȫ��������������0.5 mol�������ɣ����ȫ����������������0.25 mol�������ɡ����ݵ��ӵ�ʧ�غ����������ɵã�Cl2Ϊ0.25 mol��O2Ϊ0.125 mol�������֪��c��OH������1 mol/L��pH��14��
��ϰ��ϵ�д�
�����Ŀ
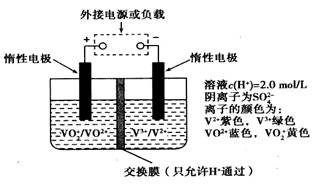
 1023���������Һ��n(H+)������0.5mol
1023���������Һ��n(H+)������0.5mol