��Ŀ����
��ҵ��������Ĺ������£�
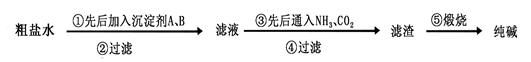
���������գ�
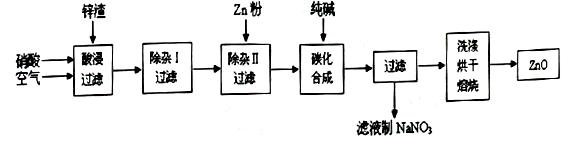
��1������ˮ������������Mg2+��Ca2+�������������A��B������(A��Դ��ʯ��Ҥ�������������B �Ļ�ѧʽΪ ��
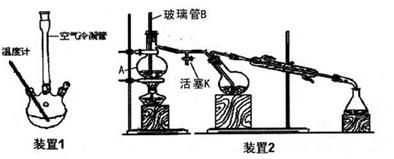
��2��ʵ����ģ������Һ�Ʊ�������װ�����£�
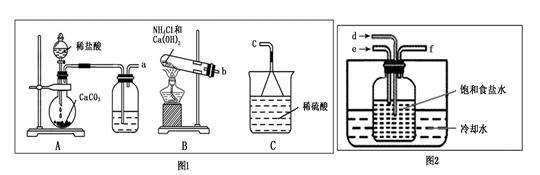
��ͼ1��װ�ú�ͼ2��װ�õ����ӷ���Ϊa�� ��b�� ��f��c��
��ͼ2���Լ�ƿ�ڷ����Ļ�ѧ��Ӧ����ʽΪ ��
��ʵ����Ҫ��ͨ���NH3����֮����ͨ��CO2���壬����ͨ���NH3�ѹ�����ʵ������� ��
��3�����������պ�Ĵ����к���δ�ֽ��̼�����ơ�ijͬѧ��ȡ�ô�����Ʒm g���ٳ�ּ������������ٱ仯ʱ�Ƶ�ʣ����������Ϊn g������Ʒ��̼���Ƶ���������Ϊ ��
��4������25���£�0.1mol/LNH3��H2O��Һ��0.1mol/LNH4Cl��Һ����������Һ�������ϲ����Һ��pH=9������˵����ȷ���� ������ţ���
a��0.1mol/L NH4Cl��Һ���Ϻ���Һ�е������ӵ��������Ŀ����ͬ
b����Ϻ����Һ�У�c(NH3��H2O)��c(Cl-)��c(NH4+)��c(OH-)��c(H+)
c���������֪��NH3��H2O�ĵ���̶ȴ���ͬŨ�ȵ�NH4Cl��ˮ��̶�
d�����ǰ������Һ��pH֮�ʹ���14

���������գ�
��1������ˮ������������Mg2+��Ca2+�������������A��B������(A��Դ��ʯ��Ҥ�������������B �Ļ�ѧʽΪ ��
��2��ʵ����ģ������Һ�Ʊ�������װ�����£�

��ͼ1��װ�ú�ͼ2��װ�õ����ӷ���Ϊa�� ��b�� ��f��c��
��ͼ2���Լ�ƿ�ڷ����Ļ�ѧ��Ӧ����ʽΪ ��
��ʵ����Ҫ��ͨ���NH3����֮����ͨ��CO2���壬����ͨ���NH3�ѹ�����ʵ������� ��
��3�����������պ�Ĵ����к���δ�ֽ��̼�����ơ�ijͬѧ��ȡ�ô�����Ʒm g���ٳ�ּ������������ٱ仯ʱ�Ƶ�ʣ����������Ϊn g������Ʒ��̼���Ƶ���������Ϊ ��
��4������25���£�0.1mol/LNH3��H2O��Һ��0.1mol/LNH4Cl��Һ����������Һ�������ϲ����Һ��pH=9������˵����ȷ���� ������ţ���
a��0.1mol/L NH4Cl��Һ���Ϻ���Һ�е������ӵ��������Ŀ����ͬ
b����Ϻ����Һ�У�c(NH3��H2O)��c(Cl-)��c(NH4+)��c(OH-)��c(H+)
c���������֪��NH3��H2O�ĵ���̶ȴ���ͬŨ�ȵ�NH4Cl��ˮ��̶�
d�����ǰ������Һ��pH֮�ʹ���14
��14�֣���1��Na2CO3��2�֣���2����d��2�֣���e��2�֣�
�� NaCl+NH3+CO2+H2O��NaHCO3��+NH4Cl��2�֣�
����պ��Ũ����IJ����������ܿ�f�����а������ɣ�˵����������������ʪ��ĺ�ɫʯ����ֽ����
�ܿ�f������ֽ������˵���������������������𰸾��÷֣���2�֣�
��3�� ��100%�����������𰸾��÷֣���2�֣� ��4�� c d��2�֣�
��100%�����������𰸾��÷֣���2�֣� ��4�� c d��2�֣�
�� NaCl+NH3+CO2+H2O��NaHCO3��+NH4Cl��2�֣�
����պ��Ũ����IJ����������ܿ�f�����а������ɣ�˵����������������ʪ��ĺ�ɫʯ����ֽ����
�ܿ�f������ֽ������˵���������������������𰸾��÷֣���2�֣�
��3��
 ��100%�����������𰸾��÷֣���2�֣� ��4�� c d��2�֣�
��100%�����������𰸾��÷֣���2�֣� ��4�� c d��2�֣������������1����������Mg2+��Ca2+�ֱ���OH����CO32����ȥ������A��Դ��ʯ��Ҥ������A����ʯ�ҡ����ڲ��������µ����ʣ���BӦ����Na2CO3��
��2����Aװ�����Ʊ�CO2��Bװ�����Ʊ������ġ����ڰ�����������ˮ��ֱ��ͨ����Һ��������������������ȷ����Ӧ����a��d��b��e��f��c��
��ͼ2���Լ�ƿ�����Ʊ�̼�����Ƶģ����Է�����Ӧ�Ļ�ѧ��Ӧ����ʽΪNaCl+NH3+CO2+H2O��NaHCO3��+NH4Cl��
�۰����Ǽ������壬��ʹʪ��ĺ�ɫʯ����ֽ������Ҳ�ܺ��Ȼ��ⷴӦ�����Ȼ�臨�ð���̣����Լ���ͨ���NH3�ѹ�����ʵ���������պ��Ũ����IJ����������ܿ�f�����а������ɣ�˵����������������ʪ��ĺ�ɫʯ����ֽ�����ܿ�f������ֽ������˵������������
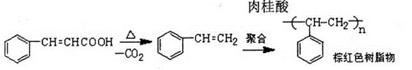
��3��̼�����Ʒֽ�ķ���ʽ��
2NaHCO3
 Na2CO3��H2O��CO2�� ��m��
Na2CO3��H2O��CO2�� ��m��2��84g 106g 62g
 g ��m��n��g
g ��m��n��g����Ʒ��̼���Ƶ�������mg��
 g��
g�� g
g���Դ�����Ʒ��̼���Ƶ���������Ϊ
 ��100%
��100%��4��0.1mol/L NH4Cl��Һ���Ϻ���Һ�е������ӵ�������ͬ������Ŀ��ͬ��a����ȷ��25���£�0.1mol/LNH3��H2O��Һ��0.1mol/LNH4Cl��Һ����������Һ�������ϲ����Һ��pH=9����Һ�¼��ԣ���˵����ˮ�ĵ���̶ȴ���NH4����ˮ��̶ȣ���˻�Ϻ����Һ��c(NH4+)��c(Cl-)��c(NH3��H2O)��c(OH-)��c(H+)��b����ȷ��c��ȷ��0.1mol/LNH3?H2O��Һ�Լ��Ժ�0.1mol/LNH4Cl��Һ�����ԣ�NH4����ˮ��̶�С�ڰ�ˮ�ĵ���̶ȣ����ǰ������Һ��pH֮�ʹ���14����d��ȷ����ѡcd��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ