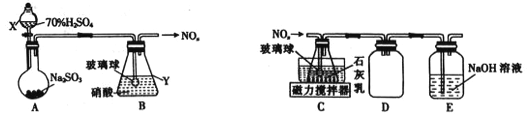
��Ŀ����
����Ŀ���ϳɾ����������オ���Ե��л��߷��Ӳ������л���ѧ�о�����Ҫ����֮һ���۴�����ϩ��(PVAc)ˮ�����ɵľ���ϩ��(PVA)�������������オ���ԣ�������������ȫ�����в����PVB���йغϳ�·����ͼ(���ַ�Ӧ�����Ͳ�����ȥ)��
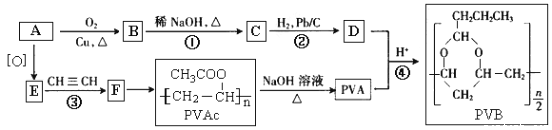
��֪��
��.AΪ����һԪ������������������ԼΪ34.8%
��.
��.![]()
��ش�
(1)C�й����ŵ�����Ϊ___________��д��C�ķ�ʽ�칹��Ľṹ��ʽ___________���÷����������___________��ԭ����ƽ�档
(2)D�뱽��ȩ��Ӧ�Ļ�ѧ����ʽΪ___________��
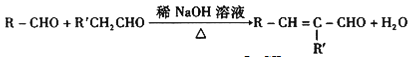
(3)���ķ�Ӧ������____________________��
(4)PVAc���ṹ��ʽΪ_______________________��
(5)д����F������ͬ�����ŵ�����ͬ���������е��������ֵĽṹ��ʽ________________________��
(6)����������Ϣ����ƺϳ�·��__________________��������Ϊԭ��(�������Լ���ѡ)�ϳ�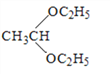
���𰸡� ̼̼˫����ȩ��  9 CH3CH2CH2CHO +
9 CH3CH2CH2CHO +![]()
![]()
![]() + H2O �ӳɷ�Ӧ
+ H2O �ӳɷ�Ӧ 
![]()
![]()
![]()


��������AΪ����һԪ����ͨʽΪCnH2n+2O����������������ԼΪ34.8%������
16/(12n+2n+2+16)��100%��34.8%�����n=2����AΪCH3CH2OH��A��������EΪCH3COOH��E����Ȳ�����ӳɷ�Ӧ����FΪCH3COOCH=CH2��F�����Ӿ۷�Ӧ�õ�PVAcΪ ������ˮ��õ�PVA��
������ˮ��õ�PVA��![]() ����A��ͭ�������������������õ�BΪCH3CHO��B������Ϣ���еķ�Ӧ�õ�CΪCH3CH=CHCHO��C������ԭ��Ӧ����DΪCH3CH2CH2CHO��D��PVA������Ϣ���еķ�Ӧ��PVB����
����A��ͭ�������������������õ�BΪCH3CHO��B������Ϣ���еķ�Ӧ�õ�CΪCH3CH=CHCHO��C������ԭ��Ӧ����DΪCH3CH2CH2CHO��D��PVA������Ϣ���еķ�Ӧ��PVB����
��1��CΪCH3CH=CHCHO��C�й����ŵ�������̼̼˫����ȩ����C�ķ�ʽ�칹��Ľṹ��ʽΪ ��ѡ��̼̼����������̼̼˫��ƽ����-CHOƽ�湲�棬����ʹ����1��Hԭ�Ӵ���ƽ���ڣ��÷����������9��ԭ�ӹ�ƽ�棻��2��D�뱽��ȩ��Ӧ�Ļ�ѧ����ʽΪ��CH3CH2CH2CHO +
��ѡ��̼̼����������̼̼˫��ƽ����-CHOƽ�湲�棬����ʹ����1��Hԭ�Ӵ���ƽ���ڣ��÷����������9��ԭ�ӹ�ƽ�棻��2��D�뱽��ȩ��Ӧ�Ļ�ѧ����ʽΪ��CH3CH2CH2CHO +![]()
![]()
![]() + H2O����3����Ӧ����CH3COOH����Ȳ�����ӳɷ�Ӧ����CH3COOCH=CH2����4��PVAc�Ľṹ��ʽΪ��
+ H2O����3����Ӧ����CH3COOH����Ȳ�����ӳɷ�Ӧ����CH3COOCH=CH2����4��PVAc�Ľṹ��ʽΪ�� ����5��FΪCH3COOCH=CH2����F������ͬ�����ŵ�ͬ���칹��Ľṹ��ʽΪ��HCOOCH=CHCH3��HCOOCH2CH=CH2��CH3OOCCH=CH2��HCOOC(CH3)=CH2����6�������鷢��ˮ�ⷴӦ�����Ҵ����Ҵ�����������������ȩ����ȩ���Ҵ���Ӧ�õ�
����5��FΪCH3COOCH=CH2����F������ͬ�����ŵ�ͬ���칹��Ľṹ��ʽΪ��HCOOCH=CHCH3��HCOOCH2CH=CH2��CH3OOCCH=CH2��HCOOC(CH3)=CH2����6�������鷢��ˮ�ⷴӦ�����Ҵ����Ҵ�����������������ȩ����ȩ���Ҵ���Ӧ�õ� ���ϳ�·������ͼΪ
���ϳ�·������ͼΪ ��
��

 ȫ�ŵ�����Ԫ�ƻ�ϵ�д�
ȫ�ŵ�����Ԫ�ƻ�ϵ�д�