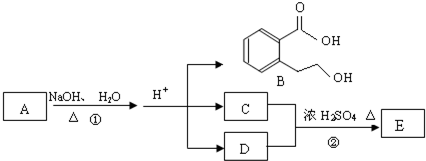
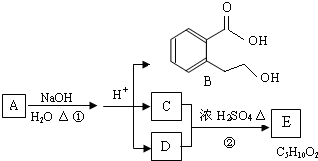
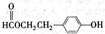��Ŀ����
A��B��C��D��E��Ϊ������ˮ�Ĺ��壬������ǵ����ӿ����У�Na+��Ca2+��Ba2+��ClO-���ֱ�ȡ���ǵ���Һ����ʵ�飬��Ҫ�������������£�
����A����Һ��ͨ�������̼���ټ���Ʒ����Һ����ɫ��ȥ��
�ڽ�B��C����Һ��ϣ����ɰ�ɫ�������ó���������E��Һ��
�۽�B��D����Һ��ϣ����ɰ�ɫ�������������������E��Һ�������ݲ�����ͬʱ���а�ɫ�������ڡ�
��A��E��Һ����ɫ��Ӧ���ʻ�ɫ��
(1)д���������ʵĻ�ѧʽ��A_________��C_________��D_________��
(2)д��A��E��Һ�У����������û���Ӧ�����ӷ���ʽΪ_____________________________
(3)��B����Һ�У�������������ϡ��E��Һ������Ҫ�����ӷ���ʽΪ__________________
(1)NaClO CaCl2��Ca(ClO)2 BaCl2��Ba(ClO)2
(2)2Al+6H+![]() 2Al3++3H2��
2Al3++3H2��
(3)H++![]()
![]()
![]()
�������������忼�ǣ��ɳ���ȷ��������ȷ������ȫȷ�����ӵķ������ɢ����������ȷ������ClO-�ɢ�ȷ������Na+�����AΪNaClO����B�ܺ�C��D���ɳ���ȷ��B�к���![]() ��C��D�к�Ca2+��Ba2+��E�к���
��C��D�к�Ca2+��Ba2+��E�к���![]() ���ڢ����������г������ɣ�˵����BaSO4��C�к�Ca2+��D�к�Ba2+��C��DΪ�������Σ����CΪCaCl2��Ca(ClO)2��DΪBaCl2��Ba(ClO)2��
���ڢ����������г������ɣ�˵����BaSO4��C�к�Ca2+��D�к�Ba2+��C��DΪ�������Σ����CΪCaCl2��Ca(ClO)2��DΪBaCl2��Ba(ClO)2��